Exobiosphere: Finding Cures from Space [Case Study]
Focusing on in-space testing and disease modeling to provide pharmaceutical companies with better data earlier in the R&D pipeline.
Exobiosphere, established in August 2024, is pioneering the pre-clinical Contract Research Organization (CRO) model for microgravity applications.
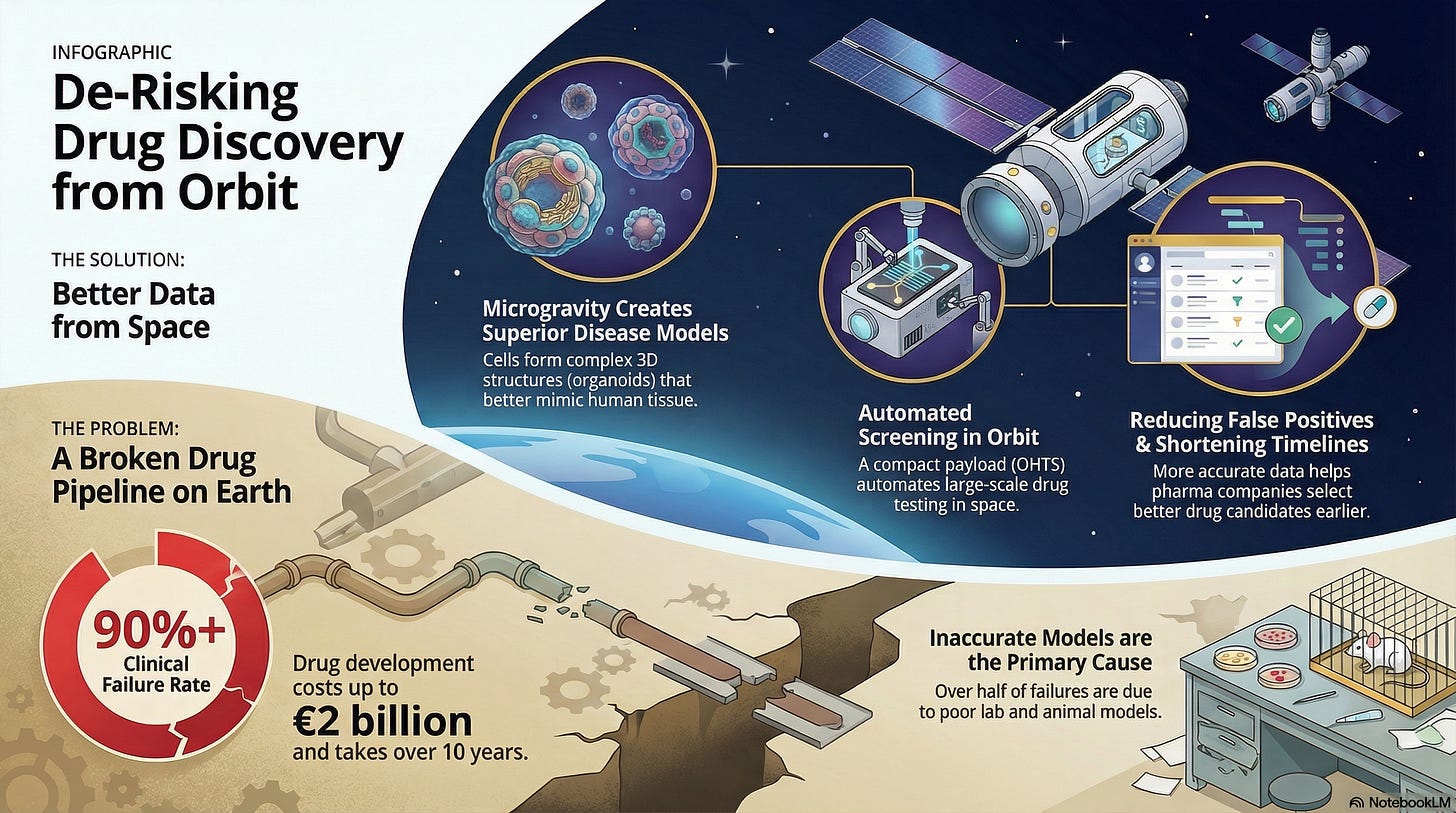
Its mission is to leverage the space environment to create superior disease models, thereby increasing the accuracy and efficiency of terrestrial drug development and reducing the 90%+ clinical failure rate that plagues the traditional pharmaceutical industry.
I. Core Value Proposition: De-Risking Pre-Clinical Trials
The company’s model is not focused on in-space manufacturing (like BioOrbit) but on in-space testing and disease modeling to provide pharmaceutical companies with better data earlier in the R&D pipeline.
The Problem Addressed (The Failure Rate): The drug development process costs up to €2 billion and takes over 10 years, with a failure rate exceeding 90%. Over half of these failures stem from inaccurate in vitro (lab-dish) and in vivo (animal) models that fail to replicate the complexity of human disease.
The Microgravity Advantage (Superior Models):
3D Tissue Growth: In microgravity, cells naturally aggregate without a scaffold, forming more complex, three-dimensional structures known as spheroids or organoids. These structures more closely mimic the human body’s native tissue and disease progression (e.g., tumor morphology) than traditional 2D cell cultures on Earth.
Enhanced Disease Modeling: This enhanced 3D growth is leveraged to create more accurate disease models for testing therapeutic compounds. This is particularly valuable for complex areas like neurodegenerative diseases (e.g., Parkinson’s) and oncology (tumor screening).
Commercial Solution: Better Pre-Clinical Data: By conducting high-throughput screening on these superior microgravity models, Exobiosphere aims to:
Reduce False Positives: Filter out ineffective compounds that appear promising in flawed Earth models.
Shorten Timelines: Accelerate pre-clinical testing, ultimately reducing the cost and duration of the R&D phase for clients.
II. Core Capabilities & IP Developed
Exobiosphere’s core IP is centered on automating the complex biological screening process within a compact, flight-qualified orbital system.
1. Orbital High-Throughput Screener (OHTS):
Core Technology: The OHTS is a compact, autonomous space payload designed to conduct automated in vitro testing on a large scale.
Function: It integrates robotic liquid handling, incubation, and imaging systems to manage and monitor thousands of individual cell assays simultaneously in microgravity.
Validation: The company completed its first parabolic flight in June 2025, successfully demonstrating its High-Throughput Screening (HTS) capabilities in weightlessness, including the operation of advanced systems like the Formulatrix Mantis liquid handler.
2. Data & Bioinformatics Platform:
IP Strategy: The service goes beyond sample return. The OHTS generates large volumes of microgravity-specific data on molecular targets and compound efficacy.
Significance: This data is useful for training the next generation of AI/ML drug discovery platforms, making the orbital service a foundational data-generation utility for AI-driven pharma R&D.
3. Ground-Based Replication and Testing:
Exobiosphere is expanding its reach by installing its biotech platform for advanced ground research at institutions like the University of Notre Dame, ensuring consistency between ground and space testing environments and facilitating client integration.
III. Venture Design & Strategic Partnerships
Exobiosphere is strategically positioned in Luxembourg, leveraging the country’s robust space and biohealth ecosystem to secure blended funding and key partnerships.
1. The Blended Funding Blueprint (Focus on Strategic Seed)
Founding & Seed Capital: It secured a €2 million Seed Round (April 2025), followed by a $1.4 million Seed Extension (November 2025). Total funding is approximately $3.6 million.
Strategic Investors: Funding was led by Expansion Ventures (focused on aerospace/defence) and included Expon Capital, Boryung Pharmaceutical (a South Korean pharma conglomerate), and Space Data Inc. (Japan).
Insight: The inclusion of Boryung Pharmaceutical is a critical validation, as it aligns the company directly with a major pharmaceutical customer and signals its commercial viability.
Accelerator and Institutional Backing:
The company joined Cedars-Sinai Accelerator+ program (November 2025), providing access to a leading US healthcare system and a further extension of capital.
Secured the LuxIMPULSE contract (March 2025) and is supported by the Luxembourg Space Agency (LSA), linking its R&D to national autonomy initiatives.
2. Strategic Partnerships for Logistics and Science
Commercial Space Station Access:
Vast Haven-1: Exobiosphere is one of the first announced tenants for the Vast Haven-1 commercial space station (launching in 2026), where it will install its OHTS device, securing access to a dedicated, long-duration orbital lab.
Space Cargo Unlimited: Partnered to use Space Cargo Unlimited’s orbital platforms (BentoBox) for high-throughput testing, further diversifying access beyond the ISS.
Logistics and Return:
ElevationSpace (Japan): Signed an MoU to enable high-frequency sample return from space, addressing the necessity of retrieving the analyzed biological samples for Earth-based analysis and further processing.
Academic and Research Collaboration:
Luxembourg Institute of Health (LIH) and LCSB: Collaborating to accelerate the development of new Parkinson’s treatments using microgravity-enhanced disease models.
IV. Execution Risk & Mitigation
Exobiosphere faces the challenge of proving that its superior orbital data can translate into statistically significant success rates in expensive, late-stage terrestrial clinical trials.
1. Technical Risk: Scalability and Sample Return Cadence
The Problem: High-throughput screening (HTS) on Earth requires continuous, rapid operation. The orbital model is constrained by launch and return opportunities, potentially creating a bottleneck in the drug discovery pipeline.
Mitigation:
Diversified Access: Mitigation is the strategy of securing contracts with multiple logistics providers and stations (Haven-1, Space Cargo Unlimited, ElevationSpace) to maximize the annual cadence of orbital missions and sample returns.
Automation Focus: The OHTS is designed for high automation, reducing the need for astronaut time and maximizing the science-per-unit-mass of the payload.
2. Translation & “Skepticism” Risk
The Problem: The conservative pharmaceutical industry requires irrefutable proof that microgravity data is predictive of clinical success and not just an interesting anomaly. They must justify adding a new, complex orbital step to their established process.
Mitigation:
Focused Proof-of-Concept: By focusing on areas where Earth models are notoriously poor (3D tumor models, neurodegeneration), Exobiosphere targets the specific pain points of pharma R&D, making the microgravity advantage functionally necessary rather than optional.
Scientific Partnerships: Strategic MoUs with key research institutions like LIH and Cedars-Sinai provide independent, institutional validation of the scientific rigor and translational value of the microgravity-derived data.
3. Competitive Risk (CRO vs. Platform)
The Problem: The competition is twofold: other general space platforms (Axiom, Nanoracks) and highly established terrestrial CROs (Evotec, Crown Bioscience).
Mitigation (Differentiation):
Exobiosphere is the world’s first dedicated Space-Based CRO, specializing in high-throughput microgravity screening. This specialization differentiates it from platform owners who only lease space, and terrestrial CROs who cannot offer the microgravity model advantage.
The Boryung investment acts as a powerful commercial shield, signaling market validation that the core service is valuable enough to warrant investment from within the pharmaceutical sector.
V. Strategic Outlook & Scaling Roadmap
Exobiosphere’s scaling roadmap is likely phased to first secure its technical and logistical viability, then to expand its therapeutic focus and achieve market dominance as the indispensable supplier of microgravity data.
Phase I: Validation & Logistical Access
Core Focus: Hardware Deployment & Proof-of-Life: Secure initial orbital presence and demonstrate OHTS reliability.
Scaling Milestones:
Successful installation and activation of the OHTS payload aboard Vast Haven-1 (2026 launch).
Execution of initial commercial research missions and successful high-frequency sample return via ElevationSpace and other partners, validating the rapid logistics chain.
Phase II: Therapeutic Expansion & CRO Integration
Core Focus: Data Translation & Client Lock-in: Move from single-target studies to supporting multi-compound screening for multiple large pharma clients.
Scaling Milestones:
Sign multi-year service contracts with at least two top-20 pharmaceutical companies.
Expand the core therapeutic focus beyond initial targets (Oncology and Neuro-degeneration) to include emerging therapeutic areas where microgravity offers a critical modeling advantage:
Inflammation
Cardiovascular Disease
Ageing
Radiation Effects
Phase III: Autonomous Scaling & Data Dominance
Core Focus: Industrialization & AI Integration: Transition to a persistent, fully industrialized orbital operation, maximizing automation to reduce per-unit-data cost.
Scaling Milestones:
Secure a dedicated, permanent module on a next-generation Commercial LEO Destination (e.g., Orbital Reef or Starlab).
Achieve full integration of the microgravity data stream into the AI/ML drug discovery platforms of partner companies, making the orbital data a prerequisite for late-stage clinical trials, and establishing Exobiosphere as a fundamental utility for biotech R&D.