High-throughput Screening (HTS) in Microgravity [Innovation]
Space-Based HTS leverages microfluidics and scaffold-free 3D cellular self-assembly to achieve higher biological fidelity than on Earth.
Orbital High-Throughput Screening (OHTS) systems are autonomous, space-grade laboratory platforms that utilize the near-weightless environment of Low Earth Orbit (LEO) to conduct thousands of parallel biochemical and cellular experiments.
While terrestrial HTS platforms rely on robotic liquid handling affected by sedimentation and convection, Space-Based HTS leverages microfluidics and scaffold-free 3D cellular self-assembly to achieve higher biological fidelity than on Earth.
In an orbital laboratory, these platforms serve two purposes: they act as a “fast-forward button” for studying aging and disease progression, and they transform the satellite into an ultra-precise bioreactor for structural biology.
I. What is Orbital High-Throughput Screening being developed for?
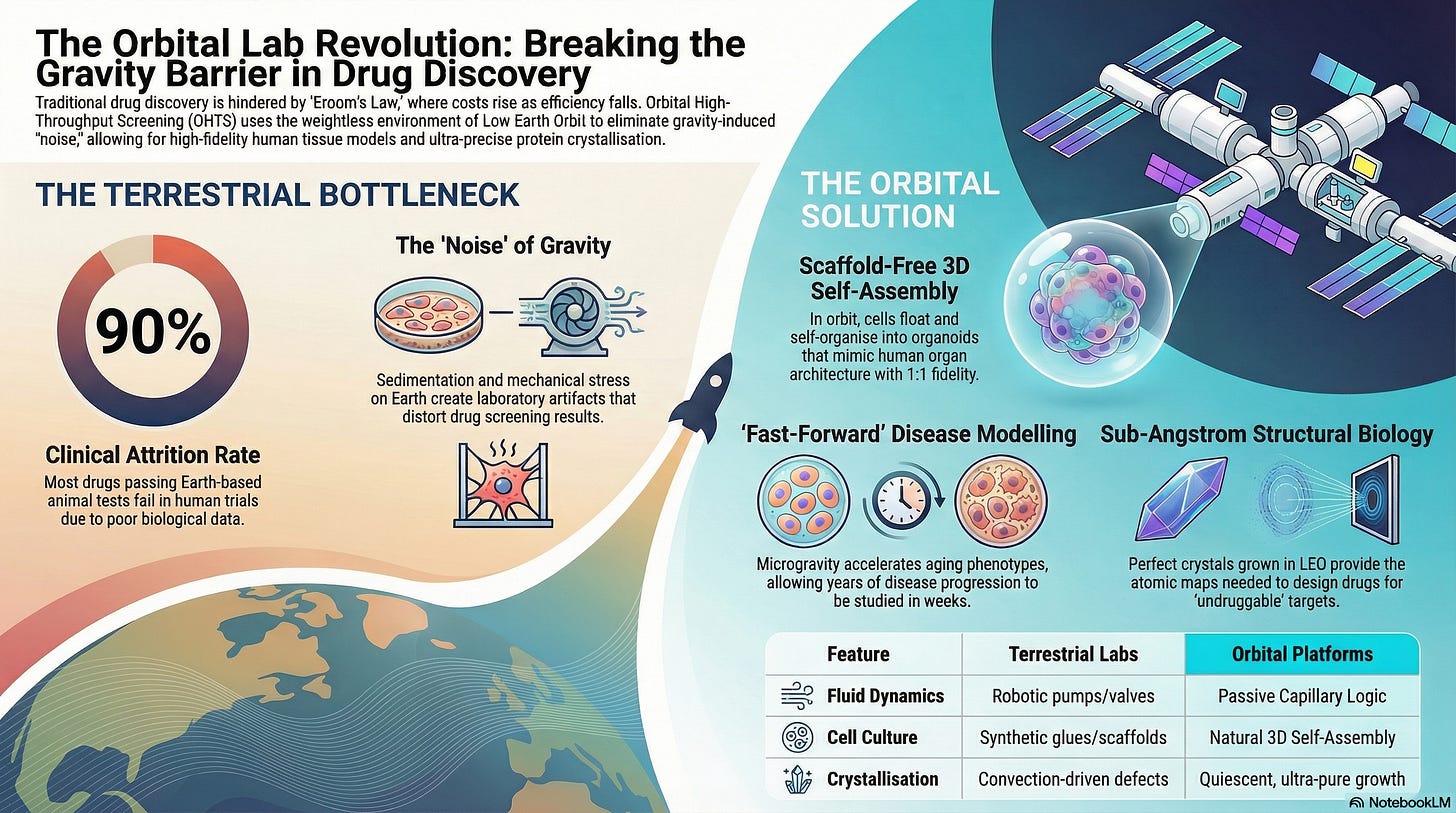
The demand for space-based screening is driven by the “Clinical Attrition Crisis,” where 90% of drugs that pass Earth-based animal tests fail in human trials. OHTS acts as the “Biological Filter,” providing more predictive data by testing drugs in an environment where human biology behaves more naturally.
1. The Strategic Level: Solving “Eroom’s Law” & Preclinical Accuracy
This tier focuses on reversing the trend of drug discovery becoming exponentially more expensive and slower. OHTS provides the “High-Resolution Truth” needed to kill failing drug candidates early.
Scaffold-Free 3D Self-Assembly: On Earth, 3D cell cultures require synthetic glues or scaffolds that alter cell behavior. In microgravity, cells float and self-organize into organoids and spheroids that mimic the architecture of human organs with 1:1 fidelity, allowing for drug screening that actually reflects human response.
Reduction of False Positives: By removing the “noise” of gravity (such as mechanical stress on cell membranes) OHTS could filter out compounds that only appear effective due to lab-induced artifacts, ensuring only the most robust “leads” proceed to human trials.
2. The Tactical Level: Accelerated Aging & “Fast-Forward” Disease Modeling
At this level, the orbital environment acts as a Biological Stressor, allowing researchers to compress years of disease progression into a few weeks of automated testing.
The “Aging-in-Orbit” Proxy: Microgravity induces rapid bone loss, muscle wasting, and immune dysfunction that mirrors human aging. OHTS platforms could screen thousands of longevity compounds or osteoporosis treatments against these accelerated phenotypes in real-time.
Neurodegenerative Mapping: Studies show that proteins associated with Alzheimer’s and Parkinson’s aggregate differently in space. OHTS systems can automate the testing of “protein-clearance” drugs in a environment where the physics of aggregation are simplified, revealing new pathways for treatment.
3. The Industrial Level: Sub-Angstrom Structural Biology
This tier focuses on the commercial necessity of mapping “undruggable” targets for the pharmaceutical industry.
Perfect-Grid Crystallization: On Earth, gravity causes convection currents that “jostle” growing protein crystals, creating defects. In the quiescent environment of LEO, OHTS can automate the growth of ultra-pure crystals with 0.2–0.3 Å better resolution, providing the “atomic map” needed to design drugs that fit perfectly into cellular receptors.
High-Concentration Biologics: OHTS is used to screen the stability of monoclonal antibodies (like those used in cancer immunotherapy). Identifying formulations that don’t “clump” in microgravity allows companies to potentially create highly concentrated doses that can be injected at home rather than administered via hospital IV.
4. The Logistical Level: Autonomous “Lab-on-a-Chip” Sovereignty
This tier addresses the engineering necessity of running a massive laboratory without a human scientist present.
Nanoliter-Scale Microfluidics: Because fluids in space are dominated by surface tension rather than weight, OHTS uses capillary-driven robotics to move nanoliter droplets. This allows the system to run 1,536-well plate experiments with 90% less reagent mass, maximizing the scientific “yield” of every kilogram launched.
AI-Integrated Tele-Science: Modern OHTS platforms, like those being deployed on commercial stations like Haven-1, integrate real-time optical sensors and AI. This allows the system to “see” a successful drug interaction and automatically pivot the next 500 tests to follow that lead.
II. Orbital High-Throughput System Types & Capabilities
Unlike the robot-arm-based labs on Earth, OHTS systems use different “fluidic architectures” to manage thousands of samples in a vacuum-sealed, weightless container.
1. Digital Microfluidic (DMF) Platforms
DMF systems move individual discrete droplets across an array of electrodes using electrowetting (controlling surface tension with electricity).
Because there are no pumps or moving valves, there is zero “dead volume” (wasted drug). It allows for the precise “merging” of a drug droplet with a cell droplet with picoliter accuracy.
Research Example: Projects by Exobiosphere and Space Tango (CubeLabs) which use “Lab-on-a-Chip” architectures to miniaturize entire HTS workflows into the size of a tissue box.
Example Application: Rare Disease Screening. Ideal for ultra-expensive orphan drugs where every microliter of the compound costs thousands of dollars.
2. Continuous-Flow “Organ-on-a-Chip” (OOC) Systems
These systems use micro-channels to provide a constant “blood flow” of nutrients to living tissue models (heart, liver, or lung) grown on a chip.
These platforms mimic the hemodynamics of the human body. In microgravity, they allow for “multi-organ” linking, where a drug’s effect on a tumor and its toxic side-effects on the liver can be measured simultaneously.
Research Example: The Tissue Chips in Space initiative (NIH/ISS National Lab), which has deployed heart and kidney chips to study tissue degradation in orbit.
Application: Safety Pharmacology. Replacing animal testing by providing a real-time map of how a drug travels through human-mimetic systems.
3. Automated Crystallization Cassettes
These are high-density storage and growth units designed specifically for structural biology, often utilizing liquid-liquid diffusion or vapor diffusion.
By precisely controlling the rate at which a protein and a salt meet in the absence of convection, these systems grow “monolithic” crystals that are often the largest and most perfect ever recorded.
Research Example: Redwire’s ADSEP (Advanced Space Experiment Processor), which uses automated cassettes to grow high-resolution crystals for companies like Eli Lilly and Merck.
Application: Structure-Based Drug Design. Providing the “3D blueprint” for the active sites of viruses and cancers that cannot be crystallized on Earth.
4. Holographic Live-Cell Imaging Systems
These “microscopes without lenses” use lasers to create 3D reconstructions of living cells as they react to drugs in real-time.
Traditional microscopy requires toxic dyes (fluorescence) that can kill cells. Holotomography (label-free imaging) allows researchers to watch the “internal density” of a cell change as it dies or heals in orbit without interfering with its biology.
Research Example: Integration of Nanolive technology into orbital incubators, allowing for 4D (3D + time) monitoring of drug efficacy.
Application: Real-Time Phenotypic Screening. Watching exactly how a cancer cell’s “skeleton” (cytoskeleton) collapses in response to a new chemotherapy agent in a weightless environment.
III. Example Business Models
These models focus on commercializing microgravity as a “Biological Utility” to secure the next generation of the global pharmaceutical economy.
1. The “Space-CRO” (Contract Research) Model
Primary Tier: The Strategic Level (Preclinical Validation) and Industrial Level.
Model: Operating as an orbital extension of a terrestrial lab. Pharmaceutical giants pay for “Orbital Campaigns” to screen their proprietary libraries. The Space-CRO manages the hardware, launch, and data retrieval, providing a “Sample-in, Data-out” service.
Innovator Opportunity: The “Orbital Lead” Filter. Offering a premium service where drugs are “vetted” in microgravity organoids to guarantee a higher success rate in Phase I human trials.
SWOT Analysis:
Strengths: Low risk for the client; clear ROI through reduced clinical attrition.
Weaknesses: High reliance on launch cadence; biological samples are time-sensitive.
Opportunities: Partnering with India’s growing CRDMO (Contract Research, Development, and Manufacturing) ecosystem.
Threats: Rapid advancement in Earth-based AI modeling reducing the perceived need for physical testing.
2. The “Orbital IP-Foundry” Model
Primary Tier: The Industrial Level (Structural Biology & Patents).
Model: Instead of charging service fees, the provider co-develops drugs with biotech firms, taking an equity stake or royalty in any IP discovered in orbit. The satellite is a “discovery engine” generating high-value patent extensions through novel crystalline structures.
Innovator Opportunity: Subcutaneous Reformulation. Using OHTS to find the one crystalline state that potentially allows a $10B/year cancer drug to be injected via syringe, effectively extending the patent life by another decade.
SWOT Analysis:
Strengths: Massive “unicorn” upside; deep alignment with pharmaceutical profit motives.
Weaknesses: High CAPEX; “all-or-nothing” reliance on the success of specific drug candidates.
Opportunities: Building a proprietary database of “Orbital Polymorphs” to license to AI drug discovery companies.
Threats: Regulatory uncertainty; the FDA has yet to finalize standards for space-manufactured biologics.
3. The “Accelerated-Omics-as-a-Service” Model
Primary Tier: The Tactical Level (Longevity & Aging).
Model: Operating a constellation of “Aging Laboratories” that provide data on how cells degrade under the stress of microgravity. Customers (longevity startups) pay for access to real-time genomic and proteomic data from their cell lines in orbit.
Innovator Opportunity: The “Digital Twin” Proxy. Using rowth in microgravity data to build AI models that can predict 10 years of human aging in seconds.
SWOT Analysis:
Strengths: Continuous revenue via data subscriptions; supports the booming longevity market.
Weaknesses: Requires high-bandwidth “downlink” for terabytes of biological imaging data.
Opportunities: Creating the “Orbital Gold Standard” for geriatric drug testing.
Threats: Human-rated commercial stations (Axiom, Sierra Space) offering “free” research slots for public PR.
4. The “Sovereign Bio-Security” Model
Primary Tier: The Tactical Level (Defense) and Logistical Level.
Model: Providing dedicated, “hardened” orbital labs for national governments to screen countermeasures against emerging pathogens or chemical threats. This model sells a “Bio-Sovereignty Hub” that is physically isolated from terrestrial interference and political borders.
Innovator Opportunity: The “Clean-Room” Satellite. Developing fully autonomous, sterile bioreactors that can synthesize and screen vaccines in orbit during a terrestrial pandemic to ensure a “clean” supply chain.
SWOT Analysis:
Strengths: Recession-proof government contracts; critical for national security infrastructure.
Weaknesses: Subject to extreme export controls (ITAR); high political sensitivity.
Opportunities: Marketing to “Neutral” states wanting an independent, space-based vaccine R&D capability.
Threats: International treaties potentially restricting “dual-use” biological research in LEO
IV. Enabling Innovations
Orbital HTS relies on Microfluidic Precision and Non-Invasive Sensing. These technologies allow satellites to manipulate biological matter with nanometer accuracy while surviving the mechanical stresses of orbit.
1. Capillary-Driven Laminar Logic
In microgravity, surface tension replaces gravity as the dominant force, allowing fluids to be moved without heavy mechanical pumps.
The Innovation: Capillary Flow Control uses the geometry of the channels themselves to “pull” fluids through the system.
SWaP Reduction: By removing traditional pumps, engineers can reduce the Size, Weight, and Power (SWaP) of the laboratory by 70%. This allows for a “massively parallel” architecture where thousands of channels operate simultaneously using only the passive physics of surface tension.
2. Lensless Digital Holographic Microscopy (DHM)
Traditional high-resolution microscopes are heavy, fragile, and have a very shallow depth of field; a major problem when cells are floating in 3D space.
The Innovation: DHM uses a laser to illuminate a sample and a high-speed sensor to capture the resulting interference pattern (the hologram).
Volumetric Imaging: Instead of focusing on a single “slice,” DHM captures the entire 3D volume of the well at once. AI algorithms then reconstruct the image, allowing researchers to track the movement and health of every cell in an organoid without ever moving a lens.
3. Acoustic Tweezers & Ultrasonic Manipulation
Handling delicate cells in a weightless environment is difficult; physical contact can trigger “stress responses” that ruin the data.
The Innovation: Acoustic Pressure Fields use ultrasonic waves to create “force cages” that can move, trap, or rotate cells without touching them.
Biological Integrity: This “no-touch” handling ensures that the cells remain in their native state. It allows OHTS systems to position spheroids perfectly within a microfluidic channel for drug exposure or imaging with zero mechanical damage.
4. Edge-AI Phenotypic Bio-Feedback
Biological experiments are unpredictable. If a cell line reacts unexpectedly to a drug, waiting for instructions from Earth (high latency) can lead to lost data.
The Innovation: Onboard Edge-AI that analyzes microscopic images in real-time to identify “hits” or biological anomalies.
Autonomous Decisioning: If the AI detects a significant cellular response, it can “auto-lock” the fluidic system to preserve that sample or trigger a follow-up dose. This ensures that the most valuable biological moments are captured even when the satellite is out of contact with ground stations.
V. Example Innovators
1. The Orbital Bioreactor & Hardware Segment (The “Foundation”)
Varda Space Industries (USA): A pioneer in autonomous pharmaceutical manufacturing. They utilize reentry capsules to return microgravity-processed drug formulations, like Ritonavir, to Earth.
Yuri Gravity (Germany): Developers of the ScienceTaxi, a fully autonomous incubator for biological samples that integrates with modular ScienceShells for diverse biotech research.
Redwire Space (USA): Operators of the BioFabrication Facility (BFF) on the ISS, specializing in 3D bioprinting human tissue and the PIL-BOX for pharmaceutical crystallization.
Space Tango (USA): Known for their CubeLab architecture and the TangoBox, a robotic laboratory hood that automates complex life science investigations in orbit.
The Exploration Company (Germany/France): Their Nyx spacecraft serves as a reusable orbital platform specifically designed to carry large-scale biotech payloads and OHTS systems.
Atmos Space Cargo (Germany): Specialized in high-capacity return capsules (Phoenix), essential for the physical retrieval of biological “hits” identified during orbital screening.
Rhodium Scientific (USA): Provides “flight-certified” biotech hardware and ensures that orbital research meets the rigorous quality standards (ISO/GLP) required by big pharma.
BioOrbit (UK): Focused on the large-scale crystallization of monoclonal antibodies in space to enable the next generation of cancer therapies.
2. The Microfluidics & “Lab-on-a-Chip” Segment (The “Gatekeepers”)
Exobiosphere (Luxembourg): Winners of the 2024 NewSpace Europe startup competition; they are aiming to launch the first dedicated Orbital High-Throughput Screener (OHTS) in 2026.
Emulate, Inc. (USA): A leader in Organ-on-a-Chip technology whose chips have been used in multiple ISS missions to study blood-brain barrier and intestinal health.
Opentrons (USA): Partnered with NVIDIA to integrate “Physical AI” into laboratory robotics, a key component for the next generation of autonomous orbital labs.
Mimetas (Netherlands): Provides the OrganoPlate platform, which enables high-throughput 3D tissue modeling without the need for traditional pumps, ideal for space-constrained payloads.
Hesperos, Inc. (USA): Known for their “Human-on-a-Chip” systems that link multiple organ types to test drug toxicity in a connected physiological environment.
CN Bio (UK): Specializes in Liver-on-a-Chip models, crucial for screening the metabolic profile and toxicity of new drug candidates during orbital missions.
3. Niche Innovators & Strategic Service Providers
Sachi Bioworks (USA): Leveraging microgravity to develop RNA-based therapeutics for neurodegenerative diseases like ALS, utilizing automated orbital screening.
Encapsulate (USA): Developing automated “tumor-on-a-chip” systems that allow for personalized cancer drug screening in the high-fidelity 3D environment of orbit.
Cedars-Sinai Board of Governors Regenerative Medicine Institute (USA): Leading research into stem cell expansion in microgravity, providing the biological “raw material” for OHTS.
Axiom Space (USA): Building a commercial space station, which will host large-scale, industrial-grade OHTS facilities for global pharmaceutical clients.
Sierra Space (USA): Their LIFE habitat and Dream Chaser spaceplane provide the pressurized volume and gentle return capability required for delicate biological screening.
LambdaVision (USA): Utilizing microgravity to manufacture protein-based artificial retinas, a process that requires the extreme layer-by-layer precision found in weightlessness.
Nanolive (Switzerland): Their Holotomography technology allows for 3D live-cell imaging without toxic dyes, integrated into orbital labs for real-time efficacy monitoring.
Microvascular Therapeutics (USA): Researching the formation of ultrasound contrast agents and drug delivery vesicles in microgravity.
Pandorum Technologies (India): A regenerative medicine startup using orbital platforms to screen and develop bio-engineered cornea and liver tissues.
VI. Potential Opportunities for New Innovators
To transition from laboratory experiments to a self-sustaining “Orbital Bio-Economy,” innovators need to solve the challenges of sample stability, autonomous decision-making, and high-fidelity return. This creates a high-value frontier for startups to build the essential “Biological Operating System” for the pharmaceutical industry of the 2030s.
1. “Biological-Handover” & Cold-Chain Logistics
Current biological samples are extremely sensitive to the temperature spikes and G-forces of launch and reentry. The next generation of OHTS requires a seamless “Life-Support Bridge” from the lab bench to the orbital slot.
The Opportunity: Developing Cryogenic “Black Boxes” and vibration-dampened launch containers that ensure 99.9% cell viability during the transition to orbit.
Innovation Focus:
Active Vitrification Systems: Developing “flash-freeze” technologies on-chip that prevent ice crystal formation without toxic cryoprotectants.
Automated Sample Transfer: Engineering the robotics for the “last mile” of space logistics; moving samples from a delivery capsule to an OHTS bioreactor without human intervention.
Example Innovators: Atmos Space Cargo (Return capsules), Rhodium Scientific (Quality assurance), Bionos (Cryo-storage).
2. Autonomous “Lab-on-a-Brain” Edge AI
As screening scales to millions of data points, the “data bottleneck” (transmitting raw video/images back to Earth) will become the primary limiting factor for orbital R&D.
The Opportunity: Building Phenotypic AI Engines that sit directly on the OHTS hardware, analyzing cell health and drug reactions in-situ and only downlinking the most critical results.
Innovation Focus:
Real-time Morphology Analysis: Training neural networks to recognize “cellular stress” or “viral inhibition” from lensless holographic data.
Adaptive Experimentation: Developing AI that can autonomously decide to increase a drug dose or change a nutrient flow-rate based on real-time biological feedback.
Example Innovators: Opentrons (AI-robotics), Nanolive (Live-cell imaging), Slingshot Aerospace (Space-edge computing).
3. “Crystal-Formulation-as-a-Service” (CFaaS)
Determining the correct “polymorph” (3D shape) of a drug is the difference between a blockbuster medicine and a biological failure.
The Opportunity: Translating raw microgravity crystallization data into Commercial Formulation Blueprints for the biologics market.
Innovation Focus:
High-Density Screening Cassettes: Creating “universal” crystallization plates that can screen 10,000 different salt and pH conditions in a single 3U CubeSat.
Atomic-Map Reconstruction: Providing the cloud infrastructure to turn orbital diffraction data into 3D molecular models for structure-based drug design.
Example Innovators: Varda Space Industries (Orbital manufacturing), BioOrbit (Antibody crystallization), Merck (Structural biology partners).
4. “Sovereign Bio-Foundries” & Orbital GMP Standards
The expansion of commercial space stations creates a market for independent biological manufacturing zones that operate under the highest international safety standards.
The Opportunity: Establishing the “Orbital GMP” (Good Manufacturing Practice); a certification layer that guarantees any drug screened or made in space meets FDA/EMA safety requirements.
Innovation Focus:
Sterile-Loop Microfluidics: Developing “self-cleaning” fluidic systems that can run for 12 months without bacterial contamination.
Regulatory Blockchain: Creating an un-falsifiable digital ledger of every temperature, pH, and chemical change a drug experienced during its orbital flight.
Example Innovators: Axiom Space (Commercial station), Sierra Space (Orbital habitats), Lonza (Terrestrial GMP experts looking upward).