Life Science Microgravity Datasets [Concept]
This piece outlines potentially high-value life science microgravity datasets that may be worth generating.
This piece outlines potentially high-value life science microgravity datasets that may be worth generating.
I. Atomic-Resolution Structural Biology (Drug Discovery)
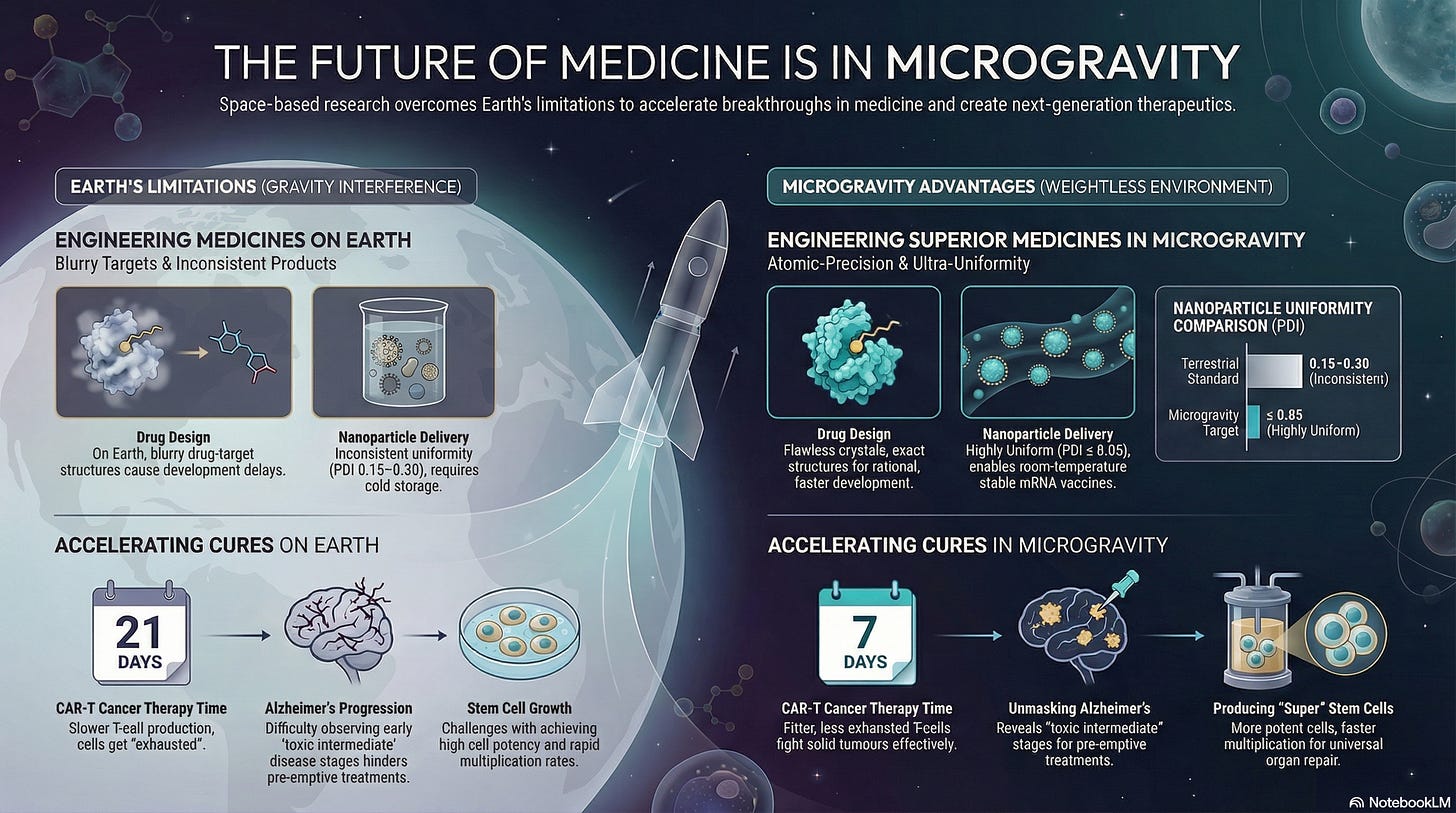
The Goal: Moving from 2.0Å (blurry/guesswork) to ≤ 1.2Å (atomic precision).
Why this is valuable vs. Earth:
On Earth, gravity-induced convection causes "lattice defects" during crystal growth. Current terrestrial datasets often lack the resolution to see hydrogen atoms or water-bridge networks.
In microgravity, growth is diffusion-limited, yielding "perfect" crystals. Moving to ≤ 1.2Å allows for Rational Drug Design, where chemists can see the exact "lock and key" fit of a molecule, potentially reducing the number of synthesis cycles from thousands to hundreds.
Evidence:
Analysis has shown that in the majority of of cases, microgravity-grown crystals achieved higher resolution than Earth controls. For example, Merck's experiments with Keytruda on the ISS produced larger, more uniform crystals that provided the high-resolution diffraction data necessary to refine its molecular structure and optimize delivery.
Researchers:
Paul Reichert (Merck Research Laboratories): A pioneer who led the Keytruda crystallization experiments on the ISS (PCG-5 and PCG-20).
Adrian Radocea & Haley Bauser (Varda Space): Published foundational work on the stability and particle size tuning of pharmaceuticals like Ritonavir in orbit.
Dataset Requirement:
Resolution: X-ray diffraction data at ≤ 1.2Å
Mosaicity: ≤ 0.1° (indicating nearly perfect internal crystal alignment).
Signal-to-Noise (I/σI): ≥ 15 in the highest resolution shell.
Pathway:
Modular, automated micro-fluidic “labs-on-a-chip” that can perform hundreds of crystallization trials in a single 10 x 10 cm (1U) unit.
Hardware:
Modular microfluidic crystallization chambers.
Key Technical Challenges:
G-Jitter: Subtle vibrations from spacecraft thrusters or life-support systems can disrupt the diffusion layer, re-introducing crystal defects.
Mechanical Re-entry Stress: Crystals are fragile; the 3g–5g loads and thermal spikes of atmospheric re-entry can fracture the lattice before synchrotron analysis.
Platform:
Varda Space W-Series Capsule. This free-flying factory provides a quiescent environment free from the larger “G-jitter” caused by astronauts on the ISS. Its autonomous re-entry is optimized to minimize the mechanical loads that would otherwise fracture a fragile 1.2Å crystal lattice.
Sample Consortium:
Pharma/Lead: Merck & Co. or Eli Lilly (Targeting insulin or antibody crystallization).
Space Infrastructure: Varda Space Industries (Autonomous re-entry) or Exobiosphere (OHTS platform).
Analysis: Diamond Light Source (UK) or ESRF (France) for ultra-bright synchrotron X-ray analysis of returned samples.
Modeling: Schrödinger, Inc. (To integrate the ≤ 1.2Å data into their physics-based drug discovery software).
II. LNP Monodispersity Benchmarks (Drug Formulation)
The Goal: Creating “Gold Standard” LNPs for stable, high-efficiency mRNA delivery.
Why this is valuable vs. Earth:
Terrestrial LNP manufacturing suffers from buoyancy-driven uneven mixing, resulting in a wide size distribution (PDI of 0.15–0.30). This lack of uniformity causes inconsistent dosing and requires ultra-cold storage.
Space-grown LNPs are “monodisperse” (perfectly uniform), which increases cell entry efficiency and could enable room-temperature stable vaccines, revolutionizing global distribution by eliminating the "ultra-cold chain" and reducing the systemic side effects caused by uneven nanoparticle sizes.
Evidence:
In the absence of gravity-driven convection, mixing occurs solely through molecular diffusion. This allow for the assembly of highly ordered, “onion-like” lamellar structures that are nearly impossible to achieve on Earth. Such precision can improve the shelf-life of mRNA therapies from ultra-cold to standard refrigerated levels.
Researchers & Organizations:
Yizhou Dong: A leading figure in LNP chemistry whose work on mRNA delivery is being benchmarked for microgravity assembly.
Dataset Requirement:
Polydispersity Index (PDI): ≤ 0.05 (Earth standard is ∼0.20)
Encapsulation Efficiency: ≥ 98% mRNA loading
Stability: Zeta potential maintenance (±5 mV) across 6 months at 4°C
Pathway:
Microfluidic mixing manifolds that use pure molecular diffusion to assemble lipid shells around mRNA strands without convective turbulence.
Hardware:
Redwire PIL-BOX (Pharmaceutical In-space Laboratory).
Key Technical Challenges:
Manifold Clogging: Without hydrostatic pressure, micro-precipitates can block 50-micron mixing channels.
Post-Assembly Ripening: Particles must be stabilized immediately to prevent merging during storage.
Platform:
Blue Origin New Shepard (Suborbital). Since LNP self-assembly occurs in seconds, the 3–5 minutes of high-quality μg provided by a suborbital flight could be sufficient. This allows for high-cadence testing and immediate recovery (within 2 hours of touchdown) for Cryo-EM analysis.
Sample Consortium:
Vaccine Lead: Moderna or BioNTech (Optimizing LNP delivery).
Hardware: Redwire Space (Specialized pharmaceutical processing payloads).
Analytics: Thermo Fisher Scientific (Providing Cryo-EM and DLS analysis of space-returned LNPs).
Flight Provider: Blue Origin (Using New Shepard for low-cost, high-cadence LNP assembly flights).
III. Accelerated Disease Modeling (Amyloid Kinetics)
The Goal: Mapping the protein-aggregation “free energy landscape” to stop Alzheimer’s before it starts.
Why this is valuable vs. Earth:
In 1g, amyloid proteins settle and clump rapidly, masking the “intermediate” stages of disease progression.
Microgravity slows this process and keeps fibrils suspended, allowing researchers to capture the toxic oligomer state; the “smoking gun” of neurodegeneration. Identifying the structure of these transient intermediates could allow for the development of pre-emptive therapeutics that neutralize toxic clusters before they form irreversible plaques, potentially treating Alzheimer's and Parkinson's years before symptoms appear.
Evidence:
JAXA experiments (2021-2024) confirmed that amyloid-β (Aβ) fibrilization is significantly slower and more “ordered” in μg. This allows for the capture of transient, high-toxicity “intermediate” states that are impossible to isolate in 1g due to rapid sedimentation and convective mixing.
Researchers:
Mitsugu Yamada (JAXA): Key investigator for the “Characterization of Amyloid Formation” project on the Kibo module.
Prof. Ben Goult (University of Liverpool): Leading researcher on the mechanical basis of Alzheimer’s and the interaction between Amyloid Precursor Protein (APP) and synaptic scaffolding.
Dataset Requirement:
Kinetics: Mapping of lag-time and growth phase at 1 nM protein concentration.
Morphology: Cryo-EM structural data of Type-1 vs Type-2 fibrils with ≤ 3Å resolution.
Inhibition Delta: 50% reduction in fibril length upon injection of candidate small molecules in-situ.
Pathway:
In-situ fluorescence imaging systems that track fibril growth across 14-30 day cycles, coupled with automated flash-freezing to “trap” transient molecular structures.
Hardware:
Holographic microscopes and in-situ flash-freezers.
Technical Challenges:
Temperature Precision: Amyloid formation is hyper-sensitive to heat; maintaining a constant 37°C across the entire payload with <0.1°C deviation is a significant engineering hurdle.
In-situ Sampling: Stopping the reaction at precise time-intervals and flash-freezing to -95°C to preserve intermediate structures.
Platform:
JAXA Kibo Module (ISS). Requires the Cell Biology Experiment Facility (CBEF) for precise thermal control (< 0.1°C variance). Long-duration observation is mandatory, making the ISS a viable home for these kinetic datasets.
Sample Consortium:
Pharma: Biogen / Eisai.
Infrastructure: JAXA (Kibo Module) / Kayser Space.
Bio-lead: University of Liverpool / ICR London.
Analytics: Oxford Nanopore (for transcriptomic stress-response).
IV. “Stemness” Expansion Maps (Regenerative Med)
The Goal: Producing “Super-Stem Cells” that don’t age during the manufacturing process.
Why this is valuable vs. Earth:
On Earth, stem cells “settle” on plastic dishes, causing mechanical stress that forces them to turn into specific tissues (differentiation) too early.
In microgravity, cells remain in a 3D suspended state that mimics the “embryonic niche.” This allows for mass-production of pluripotent cells that are more potent and have higher doubling rates than anything produced on a terrestrial factory floor. This could provide a “universal donor” source for organ repair, wound healing, and treating chronic heart disease.
Evidence:
ISS studies using induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) showed significant changes in gene expression, with 2,635 differentially expressed genes compared to Earth controls. Studies have also observed increased levels of pluripotency markers (OCT4, SOX2, NANOG) in stem cells cultured in weightless environments, effectively preserving their “potency” for longer expansion cycles.
Researchers:
Dr. Abba Zubair (Mayo Clinic): Laboratory medicine expert who has launched multiple missions to study the regenerative potential of stem cells in space for stroke and cancer.
Drs. Clive Svendsen & Arun Sharma (Cedars-Sinai): Pioneers in iPSC production in space; their research published in npj Microgravity demonstrated the first successful 3D spheroid formation and DNA transfection in orbit.
Axiom Space (Pinar Mesci, PhD): Leads the Global Head of Regenerative Medicine and Disease Modeling, coordinating with academic leads for in-space manufacturing.
Dataset Requirement:
Pluripotency Markers: ≥ 95% expression of Oct4, Sox2, and Nanog after 10 doubling cycles
Doubling Time: ≤18 hours (vs. ∼30 hours on Earth)
Transcriptomic Profile: Single-cell RNA-Seq (scRNA-seq) data comparing ≥10,000 cells to map “differentiation drift.”
Pathway:
Automated perfusion bioreactors that maintain 3D spheroid suspension while performing in-situ transcriptomic sequencing (e.g., MinION) to map “differentiation drift” in real-time.
Hardware:
Automated Perfusion Bioreactors with integrated scRNA-seq (Oxford Nanopore) for real-time transcriptomic monitoring.
Technical Challenges:
Hydrodynamic Shear: Pumping nutrients can accidentally trigger mechanosensitive pathways.
Bubble Coalescence: Managing gas exchange without “sparging” (bubbling) which kills cells in μg.
Platform:
Axiom Station Research & Manufacturing Facility (RMF). Axiom’s commercial hub is designed for biomanufacturing scale-out, providing the high-volume media exchange and “human-in-the-loop” oversight required for complex stem cell protocols.
Sample Consortium:
Clinical Lead: Mayo Clinic or Cedars-Sinai Medical Center (Existing leaders in space stem cell research).
Commercial Partner: Axiom Space (Utilizing their dedicated research hub).
Biotech: Lonza (For scaling space-based SOPs into Earth-based GMP manufacturing).
Regenerative: Sierra Space (Developing the first dedicated Space Stem Cell Institute).
V. CAR-T Persistence & Activation (Immunotherapy)
The Goal: Reducing CAR-T manufacturing from 21 days to 7 days.
Why this is valuable vs. Earth:
CAR-T therapy is currently a race against time; patients often die while their cells are being expanded in 1g labs. Furthermore, terrestrial expansion often “exhausts” the cells.
Microgravity alters the cell’s cytoskeleton (mechanotransduction), reversing exhaustion and producing “fitter” T-cells that survive longer in the patient’s body to fight solid tumors. Reducing expansion time significantly could drastically increases patient survival rates and lower manufacturing costs, potentially transforming CAR-T from a "last-resort" treatment into a primary frontline therapy for solid tumors.
Evidence:
Research has shown that microgravity can downregulate T-cell exhaustion markers like PD-1 and LAG-3. By allowing cells to “rest” from the compressive forces of gravity, the genome can undergo remodeling to resemble cells that have never been exhausted, doubling the expansion rate while maintaining superior therapeutic profile.
Researchers:
Dr. Martin Pule (University College London): Architect of the UCL CAR-T program and leader in automated manufacturing techniques for chimeric antigen receptors.
Dataset Requirement:
Exhaustion Markers: ≤ 5% expression of PD-1 and LAG-3.
Expansion Ratio: 100x expansion of CD8+ T-cells within 120 hours.
Cytokine Profile: Precise pg/mL quantification of IL-2 and IFN-gamma.
Pathway:
Closed-loop, sterile “Lab-in-a-Box” systems that automate the viral transduction and expansion of patient cells without human pipetting.
Hardware:
Microfluidic T-cell activators and CRISPR-editing manifolds.
Technical Challenges:
Automated Transduction: Inserting genes via viral vectors in an autonomous loop.
Real-time QC: Miniaturizing flow-cytometry or multiplex sensors for orbital operation.
Platform:
Space Tango CubeLabs. These standardized, automated lockers provide the sterile environment and near-real-time data downlink needed to monitor cell health and cytokine secretion profiles from the ground.
Sample Consortium:
Pharma/Cell Therapy: Novartis or Kite Pharma (Leaders in CAR-T).
Payload Specialist: Space Tango (Automated “CubeLabs” for cell culture).
Genetic Engineering: Synthego (CRISPR-editing cells before launch).
Data Science: Insitro (Machine learning to predict clinical outcomes from space-omics data).