Microgravity for Stem Cell Research [Innovation]
By removing gravity, researchers can grow massive volumes of high-purity cells that remain “stem-like” longer than their Earth-bound counterparts,
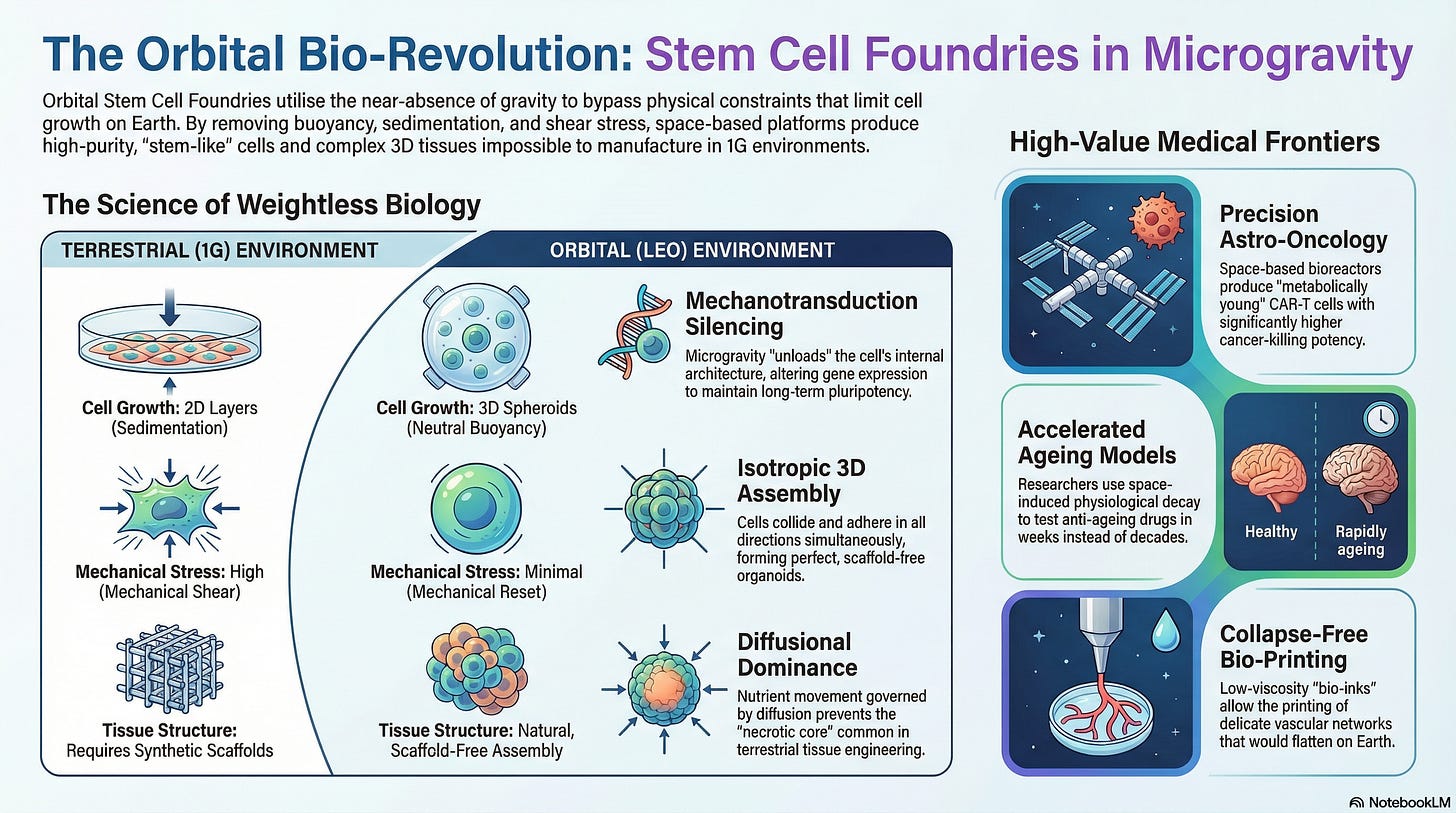
Microgravity Biomanufacturing Platforms utilize the unique physical environment of Low Earth Orbit (LEO) to bypass the “Gravitational Constraints” that limit stem cell expansion and differentiation on Earth.
While terrestrial laboratories rely on 2D plastic dishes or stirred-tank bioreactors that introduce mechanical shear stress and premature senescence, Orbital Stem Cell Foundries act as a “Biological Time Machine.”
They utilize the near-absence of buoyancy and sedimentation to maintain Pluripotency Flux and enhance the self-renewal capabilities of master cell lines.
In this research paradigm, the weightlessness of space serves as a “Structural Catalyst,” ensuring that Cellular Spheroid Uniformity and Lineage Specification Fidelity are maximized to produce the high-quality tissues required for regenerative medicine and drug discovery on Earth.
I. Where Microgravity Innovation is Most Needed?
By removing gravity, researchers can grow massive volumes of high-purity cells that remain “stem-like” longer than their Earth-bound counterparts, providing the raw material for the next generation of “off-the-shelf” cell therapies.
1. The Sub-Cellular Scale: Mechanotransduction Silencing
On Earth, gravity pulls on the cell’s cytoskeleton, triggering signaling pathways that often force a cell to mature or die prematurely. Microgravity provides a “Mechanical Reset” for the cell’s internal architecture.
Cytoskeletal Unloading: In microgravity, the actin filaments and microtubules within a stem cell relax. This “unloading” reduces the tension on the nucleus, altering gene expression patterns associated with Stemness Maintenance. This allows cells to stay in a proliferative state far longer without the risk of spontaneous, unwanted differentiation.
Nuclear Envelope Remodeling: Utilizing the lack of hydrostatic pressure to change how the DNA is packed. This creates a “Chromatin Openness” that makes the genome more receptive to reprogramming factors, effectively increasing the efficiency of creating Induced Pluripotent Stem Cells (iPSCs) from adult skin or blood cells.
2. The Tissue Scale: Scaffold-Free 3D Assembly
In a 1G environment, cells settle at the bottom of a container, forming flat layers (2D). To grow 3D tissues on Earth, scientists use synthetic scaffolds (plastic or collagen) which can interfere with cell signaling.
Isotropic Growth Environments: Without gravity, cells suspended in a liquid medium collide and adhere in all directions simultaneously. This leads to the formation of Perfect Organoids (miniature versions of hearts, brains, or livers) that more accurately mimic human anatomy than 2D culture.
Diffusional Dominance: In microgravity, the movement of nutrients and waste is governed purely by diffusion rather than convection. This creates a “Gentle Nutrient Gradient” that prevents the “Necrotic Core” (cell death in the center of a tissue) often seen in large-scale terrestrial tissue engineering.
3. The Regenerative Scale: Accelerated Aging Modeling
Spaceflight is known to induce physiological changes in astronauts that resemble rapid aging (bone loss, immune dysfunction). Researchers exploit this “Accelerated Phenotype” to study age-related diseases in weeks rather than decades.
Sarcopenia-on-a-Chip: Using microgravity to rapidly induce muscle wasting in stem-cell-derived muscle fibers. This allows pharmaceutical companies to test “Anti-Wasting” drugs in a high-throughput orbital environment, identifying candidates that can reverse frailty in the elderly on Earth.
Immunosenescence Tracking: Observing how hematopoietic (blood-forming) stem cells “age” in orbit. By understanding the “Molecular Triggers” of space-induced immune decay, scientists can develop therapies to “reboot” the immune systems of cancer patients post-chemotherapy.
4. The Macro Scale: Bio-Printing Without Slump
Traditional 3D bioprinters on Earth struggle with “structural collapse”; the bio-ink (cells and gel) often flattens under its own weight before it can solidify.
Low-Viscosity Bio-Inking: In orbit, researchers can use much “thinner” and more biological bio-inks because the printed structure will not collapse. This allows for the printing of complex, delicate vascular networks (capillaries) that are currently impossible to manufacture on Earth.
Cryogenic Preservation Integration: Developing “Orbital-to-Earth” cold chains where complex 3D-printed tissues are flash-frozen in microgravity to preserve their delicate internal architecture, ensuring they survive the return trip through the atmosphere for eventual transplant into patients.
II. Potentially High-Value Research Targets
1. Neuro-Regenerative Medicine (The “Neural Mapping” Frontier)
Some valuable breakthroughs involve reversing the damage of neurodegenerative diseases. On Earth, brain cells (neurons) are notoriously difficult to grow into complex, functional networks due to gravity-induced sedimentation.
Alzheimer’s & Parkinson’s Disease Modeling:
Creating high-fidelity Midbrain Organoids.
In microgravity, dopaminergic neurons can form the intricate, three-dimensional axonal connections that characterize the human “Substantia Nigra.”
These space-grown tissues serve as a superior platform for testing small-molecule drugs that could stop protein misfolding (Tau/Amyloid-beta) before symptoms appear.
Myelin Sheath Repair:
Engineering Oligodendrocyte Progenitor Cells (OPCs).
Microgravity appears to accelerate the maturation of the cells responsible for insulating nerve fibers.
This research aims to develop “Injectable Progenitors” that can migrate to lesions in the central nervous system to treat Multiple Sclerosis.
2. Hematopoietic & Immunotherapy (The “Astro-Oncology” Scale)
Microgravity is being used to “re-educate” the immune system. By removing the physical stressors of Earth, researchers can produce more aggressive and precise cancer-fighting cells.
Ex Vivo T-Cell Expansion:
Generating high-potency CAR-T Cells.
Current Earth-based manufacturing often leads to “T-cell exhaustion,” where the cells become too tired to fight the tumor.
Space-based bioreactors maintain the “Metabolic Youth” of these cells, resulting in a therapeutic product with a significantly higher Cytotoxic Kill Rate against solid tumors.
Leukemia Stem Cell Quiescence:
Identifying the “Shielding Mechanisms” of cancer stem cells.
In microgravity, certain cancer cells become more sensitive to chemotherapy.
By studying this “Vulnerability Window,” researchers could identify new genetic targets to prevent cancer relapse on Earth.
3. Orthopedic & Cardiac Bio-Manufacturing (Engineering Resilience)
The lack of sedimentation allows for the assembly of dense, weight-bearing tissues that are impossible to knit together in a 1G environment.
Hyper-Pure Chondrocyte Scaffolding:
Growing Articular Cartilage for joint replacement.
On Earth, lab-grown cartilage is often “patchy” and weak.
Microgravity ensures a perfectly uniform distribution of the extracellular matrix (Type II Collagen), creating “Biological Implants” that can withstand the mechanical loads of human walking without the need for synthetic resins.
Cardiac Patch Vascularization:
Printing Vascularized Myocardium.
One of the greatest challenges in heart-tissue engineering is getting blood vessels to grow deep into the tissue.
The “Isotropic Diffusion” of microgravity allows endothelial cells (vessel-formers) to weave perfectly through cardiac muscle, creating “Live Patches” that can be grafted onto a scarred heart after a myocardial infarction.
IV. Example Business Models
These models could transition space-based stem cell research from a purely scientific pursuit into a high-value industrial pipeline, addressing the multi-billion dollar markets for regenerative medicine and drug development.
1. The “Stemness-as-a-Service” (StaaS) Model
Primary Tier: The Mechanotransduction Scale (Sub-Cellular) and Scaffold-Free Assembly (Tissue Scale).
Model: Partnering with major pharmaceutical and biotech firms (e.g., Lonza, Pfizer) to act as their “Orbital Manufacturing Arm.” Instead of the client managing complex orbital logistics, the provider operates automated bioreactors to expand high-purity stem cell batches or grow complex organoids. Revenue is generated via Mission-Based Processing Fees and a “Quality Premium” for cells that maintain higher potency than Earth-grown equivalents.
Innovator Opportunity: The Digital Bio-Twin Library. Using real-time sensor data from orbital bioreactors to create AI models that predict how specific patient cell lines will behave in microgravity, allowing for “Precision Simulation” before a sample ever leaves Earth.
SWOT Analysis:
Strengths: Solves the “Scalability Crisis” for autologous therapies; provides a unique, defensible IP through microgravity-specific cell signatures.
Weaknesses: High initial “Cost-per-Batch” compared to terrestrial stirred-tank bioreactors.
Opportunities: Expansion into the Longevity Market by producing “rejuvenated” stem cell banks for healthy aging.
Threats: Rapid improvements in terrestrial 3D-bioprinting or magnetic levitation systems that simulate weightlessness.
2. The “Orbital Organoid Foundry” (OOF) Model
Primary Tier: The Tissue Scale (3D Assembly) and Regenerative Scale (Accelerated Aging).
Model: Specializing in the production of high-fidelity human organoids (heart, brain, liver) for pre-clinical drug testing. Pharmaceutical companies pay for “Test-Ready Tissues” that have been aged in orbit to mimic chronic diseases like Alzheimer’s or Sarcopenia. This model acts as a “Failure Filter,” helping companies identify toxic or ineffective drugs years earlier than terrestrial trials.
Innovator Opportunity: In-Situ Multi-Omic Screening. Integrating automated gene-sequencing and metabolic profiling within the space station or satellite, delivering a complete “Biological Data Package” to the client alongside the physical tissue.
SWOT Analysis:
Strengths: High demand for more accurate human models to replace animal testing; “accelerated aging” is a unique orbital value proposition.
Weaknesses: Extremely delicate “Cold Chain” logistics required for returning live tissues to Earth without damage.
Opportunities: Becoming the “FDA-Gold Standard” for high-resolution toxicology testing.
Threats: Development of highly complex “Organ-on-a-Chip” systems that can simulate disease without the need for microgravity.
V. Enabling Technologies
Orbital Stem Cell Foundries rely on Bio-Oscillation Bioreactors and Shear-Mitigating Fluidic Architectures. These technologies allow the cell to exist in a state of “Mechanical Suspension,” distinguishing between the life-sustaining nutrient flow required for survival and the subtle gravitational unloading necessary to drive cellular innovation.
1. Acoustic Levitation & Electromagnetic Positioning
Standard terrestrial bioreactors use mechanical impellers (blades) to keep cells from settling, which creates “High-Shear Stress” that can rupture delicate stem cell membranes.
The Innovation:
Ultrasonic Standing Wave Nodes use high-frequency sound waves to “trap” clusters of stem cells in mid-air (or mid-liquid) without physical contact.
This allows for Isotropic Nutrient Access, where every side of a 3D organoid receives equal oxygen and growth factors, preventing the “asymmetric maturation” seen in 1G environments.
Active Diamagnetic Levitation:
Utilizing superconducting magnets to provide a “Fine-Tuning” force.
This mimics a “Variable Gravity” environment (e.g., 0.16G for Moon or 0.38G for Mars), allowing researchers to calibrate the exact level of mechanical stress needed to trigger specific regenerative pathways.
2. Capillary-Driven “Synthetic Vasculature”
On Earth, pumping fluids through dense tissue requires high pressure, which can collapse the fragile “vessel-like” structures forming within an organoid.
The Innovation:
Biomimetic Wicking Channels utilize the absence of buoyancy to move media through surface tension alone.
Instead of high-pressure pumps, these systems use engineered “Micro-Grooves” that pull nutrients deep into the center of a 3D tissue mass.
Metabolic Feedback Loops:
These systems integrate Graphene-Based Nanosensors that detect real-time changes in pH, glucose, and lactate.
The system then automatically adjusts the “Wicking Rate,” ensuring the stem cells are never starved of energy during rapid expansion phases.
3. Soft-Capture Reentry & Vibration Dampening
The transition from the weightlessness of orbit to the high-G forces of atmospheric reentry can trigger “Stress-Induced Apoptosis” (cell death), undoing weeks of orbital growth.
The Innovation:
Non-Newtonian Fluid Dampeners provide a “Biological Shock-Absorber.”
By surrounding the cell canisters with shear-thickening fluids (which stay liquid during orbit but turn solid/gel-like during the vibration of reentry), the delicate 3D architecture of the organoids is physically locked in place.
Automated Cryo-Vitrification:
Before the descent begins, some platforms utilize Rapid-Flash Cooling.
By bypassing the formation of ice crystals (vitrification), the cells are “paused” in their orbital state.
This ensures that the “Stemness” captured in LEO is perfectly preserved until the capsule is opened in a terrestrial lab.
4. Gas-Permeable Fluoropolymer Housing
Traditional plastics can “outgas” volatile organic compounds (VOCs) that act as endocrine disruptors, potentially “confusing” the stem cell’s differentiation signals.
The Innovation:
Nanoporous PTFE-Composite Membranes are engineered to be “chemically inert” while allowing for high-efficiency gas exchange.
These materials allow the culture to “breathe” O2 and CO2 with zero risk of chemical leaching from the container walls, even during the thermal cycles of an orbital sunrise.
Atmospheric “Scrubbing” Modules:
The housing includes a Molecular Sieve made of specialized zeolites.
This filters out the “Spacecraft Smell” (trace ammonia and metallic ions) common in the ISS environment, ensuring the stem cells are grown in a “Pristine Biological Void,” free from terrestrial and orbital pollutants.
VI. Example Innovators
1. The Cellular Expansion & Biomanufacturing Segment (The “Engine”)
Dr. Arun Sharma (Cedars-Sinai Medical Center, USA): Director of the Center for Space Medicine Research; he pioneered the long-duration cardiac stem cell experiments on the ISS and successfully reprogramed human cells into iPSCs in orbit.
Dr. Dhruv Sareen (Cedars-Sinai Biomanufacturing Center, USA): An innovator in automated orbital cell culture; he leads the development of shoebox-sized automated “foundries” for mass-producing stem cells in LEO.
Dr. Catriona Jamieson (UC San Diego, USA): A leader in “Astro-Oncology”; she utilizes the Sanford Stem Cell Orbital Research Laboratory to study how blood cancers evolve in microgravity, identifying new therapeutic targets for leukemia.
Kenneth Savin, Ph.D. (Redwire Space, USA): He directs the commercialization of the BioFabrication Facility (BFF), the first 3D-bioprinter capable of printing human tissue patches in space.
Dr. Clive Svendsen (Regenerative Medicine Institute, USA): A world-renowned neuroscientist focusing on “Patient-in-Orbit” models; his work involves sending patient-specific stem cells to the ISS to develop personalized treatments for ALS and Parkinson’s.
Dr. Miria Ricchetti (Institut Pasteur, France): Principal Investigator of the “Cerebral Ageing” project; her team successfully cultured the first live human brain organoids to return from orbit, providing insights into accelerated molecular aging.
Dr. Alysson Muotri (UC San Diego, USA): A pioneer in “Space Organoids”; he uses brain-in-a-dish models on the ISS to study neurological network activity and neurodevelopmental disorders like Autism and Alzheimer’s.
Valentina Fossati, Ph.D. (New York Stem Cell Foundation, USA): Specializes in orbital neuro-immunology; she investigates how the immune cells of the brain (microglia) drive inflammation in Multiple Sclerosis patients under microgravity stress.
2. The Physiological & Molecular Research Segment (The “Insight”)
Dr. Sonja Schrepfer (Sana Biotechnology / Cedars-Sinai, USA): An expert in “Hypo-immunogenic” cells; her research focuses on how microgravity alters the immune system’s recognition of stem cell grafts.
Dr. Egle Cekanaviciute (NASA Ames Research Center, USA): Investigates the impact of space radiation and weightlessness on the blood-brain barrier using complex “Organ-on-a-Chip” models.
Dr. Marie Denise Roggan (German Aerospace Center - DLR, Germany): Researches astrocyte behavior and DNA damage repair mechanisms in response to the high-LET radiation environment of deep space.
Dr. Davide Marotta (NYSCF / National Stem Cell Foundation, USA): A key researcher in 3D-disease modeling who bridged the gap between patient-derived cells and orbital hardware logistics for neurodegeneration studies.
Dr. Joseph Wu (Stanford Cardiovascular Institute, USA): A leading cardiologist who utilizes spaceflight to study the functional and transcriptomic changes in human heart cells, aiming to prevent “Spaceflight-Induced Heart Atrophy.”
Dr. Marjana Tomic-Canic (University of Miami, USA): An innovator in wound healing; her team sent human skin cells to the ISS to study how microgravity can unlock novel regenerative pathways for chronic terrestrial wounds.
Dr. Irena Pastar (Miller School of Medicine, USA): Co-investigator in space-based tissue repair research, focusing on the genomic signals that trigger rapid skin cell regeneration in weightless environments.
3. Niche Innovators & Strategic Service Providers
RoosterBio (USA): A primary industrial partner providing standardized, “fit-for-space” Mesenchymal Stem Cell (MSC) banks and media systems for orbital expansion.
Lonza Group (Switzerland): A terrestrial biomanufacturing giant that provides the specialized media and “stemness-maintaining” reagents used in many orbital bioreactors.
Sanford Stem Cell Clinical Center (USA): The clinical hub responsible for translating “Space-Proven” stem cell discoveries into FDA-approved human clinical trials on Earth.
VII. Potential Opportunities for New Innovators
To transition from “one-off” orbital experiments to a standardized Space-to-Clinic Industrial Pipeline, innovators must solve the challenges of in-situ biological monitoring, high-cadence sample recovery, and AI-driven stability tracking. This creates a high-value frontier for startups to build the “Biotic Infrastructure” for the regenerative medicine of the 2030s.
1. In-Situ “Bio-Sensing” & AI Morphological Tracking
Current stem cell research often relies on “post-flight” analysis; researchers don’t know the quality of their organoids until they land.
The Opportunity: Developing “Smart-Bioreactors” that use real-time computer vision and edge-AI to monitor cellular health and differentiation markers while in orbit.
Innovation Focus:
Multi-Parametric Fluorescence Imaging: Miniaturized microscopes that can detect Lineage-Specific Reporters (e.g., green-glowing heart cells) to verify successful differentiation in real-time.
Edge-AI Morphology Sorting: Training algorithms to identify “phenotypic outliers” (cells that expand faster or show superior structural integrity) allowing for the autonomous selection of the highest-quality batches for return.
Digital-Twin Biomanufacturing: Creating software that uses orbital sensor data to predict the terrestrial performance of a tissue patch, allowing clinics to prepare for a patient’s surgery before the cells even de-orbit.
Example Innovators: Yuri Gravity (Modular science labs), Space Tango (Automated CubeLabs), Ozette Technologies (AI-driven immune monitoring).
2. High-Cadence “Cold-Chain” Recovery Logistics
The primary bottleneck for orbital biomanufacturing is the lack of dedicated, low-cost recovery vehicles that can return delicate biological samples without the “G-Shock” of a standard ocean splashdown.
The Opportunity: Engineering “Pico-Recovery” capsules (sub-100 kg) optimized specifically for the “Live Return” of stem cell products.
Innovation Focus:
Active-Vibration Cancellation: Capsules utilizing magnetic or fluidic dampening to protect delicate 3D organoid structures from the intense mechanical resonance of atmospheric friction.
Precision “Dry-Land” Recovery: Utilizing GPS-guided para-gliders to land biological canisters directly at clinical processing centers (Hospitals-in-Space Hubs), bypassing the delays of naval recovery.
Cryo-Stasis Capsules: Reentry vehicles with integrated liquid nitrogen or thermoelectric cooling systems that maintain a strict -80C or -196C “Cold Chain” through the 1,500C heat of reentry.
Example Innovators: Genesis Space Flight Laboratories (GEN Capsules), Varda Space Industries (Automated space factories), Atmos Space Cargo (Low-G return vehicles).
3. “Zero-Drift” Genomic & Epigenetic Stability Tracking
To be clinically viable, space-manufactured cells must remain genetically stable and not “drift” toward oncogenic (cancer-forming) states due to cosmic radiation.
The Opportunity: Building “Epigenetic Stability Monitors” and tools that verify the safety of space-grown therapies.
Innovation Focus:
On-Orbit Nanopore Sequencing: Integrating miniaturized gene-sequencers (like Oxford Nanopore) directly into the bioreactor to perform “Quality Control Checks” on the DNA code before de-orbiting.
Epigenetic “Locking” Buffers: Developing biochemical media that “freeze” the beneficial methylation patterns induced by microgravity, ensuring the cells retain their “youthful” orbital state once back on Earth.
Active Dosimetry-Bioreactor Integration: Creating sub-millimeter radiation maps that correlate specific cosmic ray “hits” with cellular mutations, allowing for the automated culling of damaged samples.
Example Innovators: BioServe Space Technologies (Hardware), Nanobiosym (Real-time gene detection), Oxford Nanopore (Miniaturized sequencing).
4. The “Tissue-as-a-Service” (TaaS) Digital Marketplace
The future of space-based medicine is both the physical cell and the unique “Omic” data generated in the weightless environment.
The Opportunity: Creating a “Cloud-Biology” platform where pharmaceutical companies can license the “Digital Profiles” of space-aged tissues for drug discovery.
Innovation Focus:
Standardized Organoid Libraries: Selling digital access to the transcriptomic and proteomic maps of thousands of “Space-Aged” tissues for rapid toxicity screening.
Blockchain Bio-Provenance: Using decentralized ledgers to track a cell line’s “Flight Heritage,” ensuring that the safety and purity records of a “Space-Origin” therapy are immutable for FDA certification.
Biosecurity “Vault” Subscriptions: Offering patients a recurring service to store their personal stem cell “backups” in a radiation-hardened, orbital facility for future regenerative needs.
Example Innovators: Satsearch (Space supply marketplace), D-Orbit (Orbital logistics), Sanford Stem Cell Institute (Clinical translation).