Microgravity Pre-Clinical Oncology Workflows [Strategy]
While Conventional workflows remain the bedrock of standard drug development, the emergence of Hybrid and Microgravity-Maximal strategies could fundamentally alter how we define pre-clinical success.
While Conventional workflows remain the bedrock of standard drug development, the emergence of Hybrid and Microgravity-Maximal strategies could fundamentally alter how we define “pre-clinical success.”
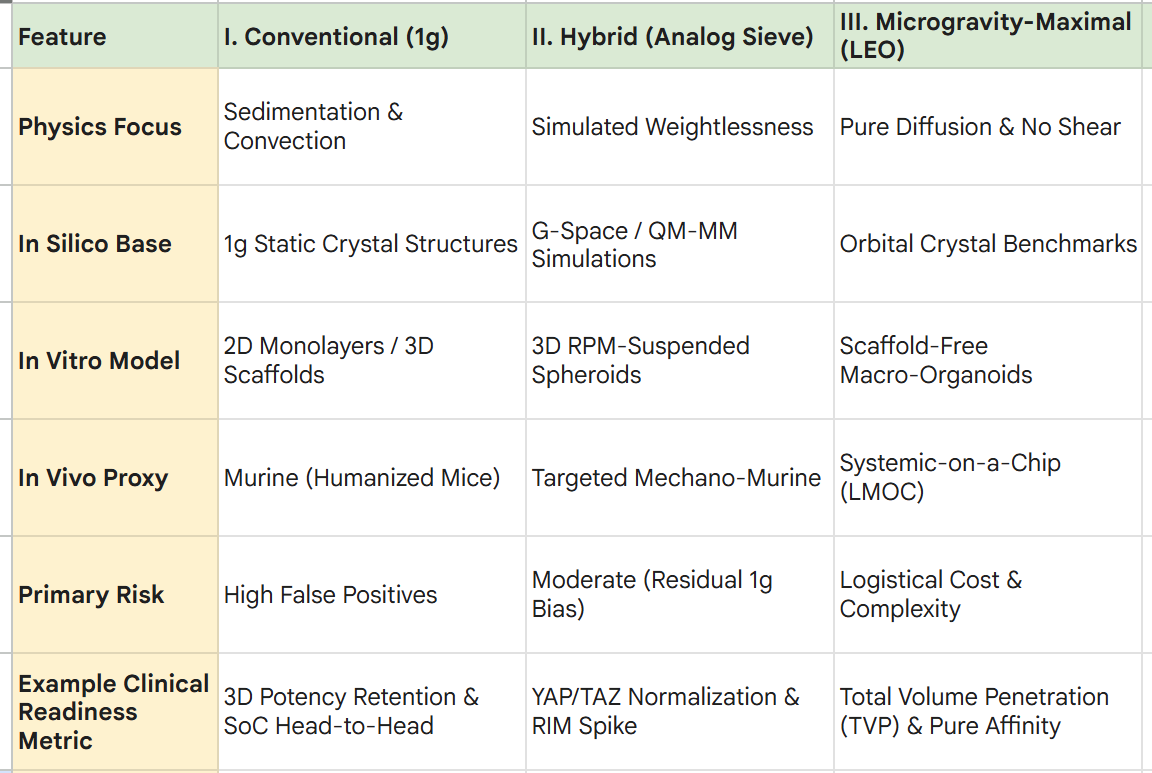
I. The Conventional Workflow (Earth Limitations)
1. In Silico (“Static Crystal” Constraint)
Traditional computational modeling relies on protein structures derived from X-ray crystallography or Cryo-EM performed in 1g.
The Limitation:
These structures represent a “compressed” state. On Earth, the internal scaffold of a protein is under the constant pull of gravity and the mechanical tension of the cell’s cytoskeleton.
The Consequence:
Molecular docking simulations often miss “cryptic pockets” that only open when a protein is in a relaxed, weightless state.
This leads to False Negatives, where potentially life-saving drugs are discarded because they don’t “fit” the distorted 1g crystal structure.
Terrestrial Strategies & Limits:
Strategy:
Researchers employ Molecular Dynamics (MD) simulations and Enhanced Sampling (e.g., Metadynamics) to computationally “tug” on protein structures, attempting to simulate conformational flexibility and “force-open” hidden pockets.
The Limit:
These simulations are computationally expensive and rely on mathematical approximations of force.
Without a true microgravity benchmark, it is nearly impossible to know if the “simulated relaxation” accurately reflects the biological reality of a weightless cell, often leading to “hallucinated” pockets that do not exist in vivo.
2. In Vitro (“Flattened Biology” Problem)
The transition to “wet lab” testing on Earth introduces physical artifacts that distort biological truth.
The Limitation:
Cells are grown on flat plastic surfaces (2D) or as small, gravity-stunted spheroids (3D).
This forces a 3D biological entity into a “pancake” or a compressed sphere, fundamentally altering gene expression and surface receptor density.
The Consequence:
Drugs are validated against a distorted cellular architecture.
A drug might show high efficacy against a flattened cell but fail to penetrate the dense, pressurized extracellular matrix (ECM) of a real human tumor.
Terrestrial Strategies & Limits:
Strategy:
Utilization of Hydrogels and Synthetic Scaffolds (e.g., Matrigel) to provide a 3D structural “cage” that mimics the body’s natural environment.
The Limit:
These scaffolds introduce non-biological variables.
The plastic or protein-mesh scaffolds create artificial diffusion barriers and “stiffening” effects that do not exist in the fluid-like, scaffold-free environment of microgravity, often masking the drug’s true penetration speed.
3. In Vivo (“Murine Proxy” Gap)
The final pre-clinical step usually involves “humanized” mice.
The Limitation:
A mouse is a 1g organism. Its circulatory system, interstitial fluid pressure, and lymphatic drainage are all evolved to function under Earth’s gravity.
The Consequence:
When we test a human CAR-T cell in a mouse, we observe interactions in a high-convection, high-sedimentation environment.
The “trafficking” of the T-cell is influenced by the mouse’s gravity-driven drainage, which does not replicate the diffusion-dominated microenvironment of a dense human tumor.
Terrestrial Strategies & Limits:
Strategy:
Microfluidic Organ-on-a-Chip (OOC) systems are used to simulate human-like flow and pressure without using animals.
The Limit:
In 1g, OOC systems suffer from channel clogging and sedimentation.
To keep cells suspended, researchers must use high-flow pumps that introduce “fluid shear stress,” which can accidentally trigger inflammatory pathways in the cells, creating “false signals” that would not occur in the calm, low-shear environment of space.
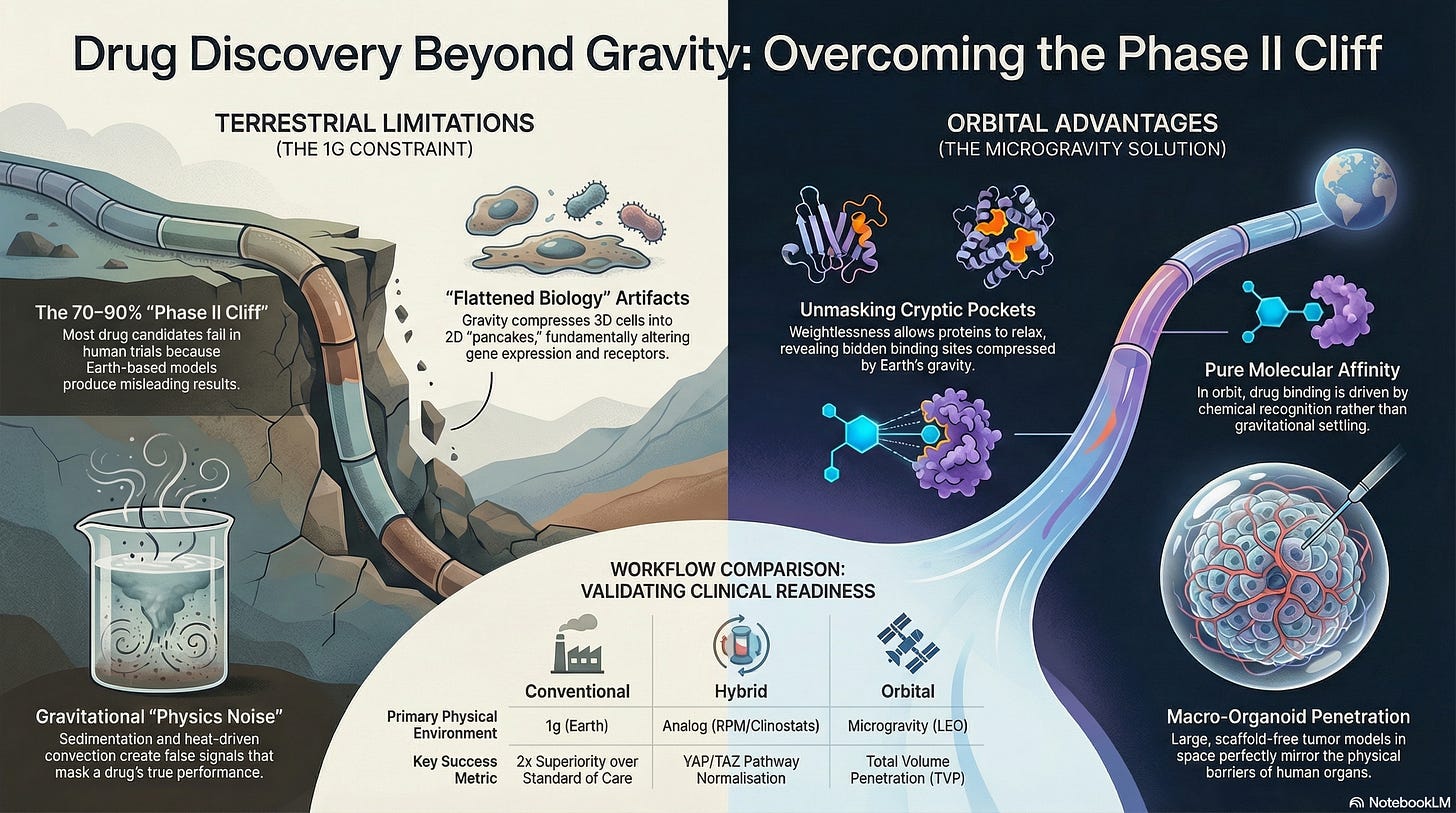
4. Workflow Risk (“Phase II Cliff”)
The greatest risk is the False Positive. A drug candidate looks spectacular in a 2D dish because it settled perfectly onto the receptors (Sedimentation) and moved quickly through the media (Convection).
However, when that drug enters a human patient, it encounters:
High Interstitial Pressure: The drug can’t “fall” into the tumor.
Diffusion-Limited Transport: There are no convection currents in the center of a dense pancreatic tumor.
Mechanical Resistance: The tumor stroma is a vertical wall, not a flat plastic surface.
5. Clinical Readiness
Despite its inherent limitations and the “Physics Noise” of Earth, the Conventional Workflow still produces the majority of candidates for human trials. To maximize the odds of surviving the “Phase II Cliff,” researchers look for specific, high-stress indicators that go beyond simple 2D efficacy. These are the results that allow a program to proceed with “cautious optimism” into the clinic.
A. Multi-Model “Infiltration Persistence”
Since 1g sedimentation often creates a False Positive for drug binding, the most reliable result is one that holds up across variable mechanical environments.
The Result:
The drug shows high potency in 2D monolayers, and retains at least 40% efficacy in dense 3D hydrogel-scaffolded spheroids.
Clinical Significance:
This “Potency Retention” suggests that the molecule’s chemical affinity is strong enough to overcome at least some physical barriers.
It indicates that the drug isn’t just “falling” onto cells but is actively navigating the interstitial space.
B. The “Stress-Induced” Toxicity Benchmark
Because in vitro cells on plastic are often “unusually happy,” they don’t reflect the stressed environment of a human body.
The Result:
The drug is tested on cells under hypoxic (low oxygen) and acidic conditions (mimicking a tumor’s core) and maintains its therapeutic window.
Clinical Significance:
Validating that the drug’s mechanism isn’t neutralized by the harsh metabolic environment of a real tumor is a critical green light for human dosing.
It reduces the risk of the drug becoming “inert” once it leaves the controlled lab incubator.
C. Orthotopic Murine “Signal-to-Noise”
Standard subcutaneous (under the skin) mouse models are notoriously poor predictors. The conventional gold standard is the Orthotopic Model, where human tumor cells are implanted directly into the corresponding organ (e.g., human breast cancer in a mouse mammary fat pad).
The Result:
Significant tumor regression in an orthotopic model with biomarker evidence of the drug reaching the center of the mass.
Clinical Significance:
While the mouse is still a 1g proxy, an orthotopic model forces the drug to navigate organ-specific blood flow and tissue density. If the drug works here, it provides the “Biological Proof of Concept” required by the FDA/EMA for an IND filing.
D. Comparative “Standard of Care” (SoC) Head-to-Head
In 1g research, the absolute value of a drug is less important than its relative value.
The Result:
The candidate drug consistently outperforms the current “Standard of Care” (e.g., Paclitaxel or Gemcitabine) by a factor of 2x in both in vitro 3D models and in vivo mouse models.
Clinical Significance:
This “Super-Superiority” creates a buffer. Even if Earth’s physics noise inflated the results by 30%, the drug is still likely to be more effective than existing treatments in a human patient.
Even with these results, a Conventional Workflow candidate still faces a 70-90% failure rate in Phase II because it has never been tested in a diffusion-dominated, scaffold-free environment.
These terrestrial results prove the drug is “chemically potent,” but they cannot prove it is “physically capable” of navigating a human body as well as an orbital-validated candidate.
II. A Hybrid Workflow (The “Analog Sieve”)
Focus: Integrating computational biology (G-Space) and ground-based analogs (Litegrav) to filter hypotheses and maximize the probability of success (PoS) before committing to 1g animal trials.
The Hybrid Workflow serves as a middle ground. It acknowledges that while orbital slots are scarce, terrestrial “1g-only” logic is flawed.
By using a tiered “sieve” approach, researchers can potentially capture high-value biological signals (specifically those related to mechanotransduction and pure molecular affinity) that can be drowned out by Earth’s gravitational noise.
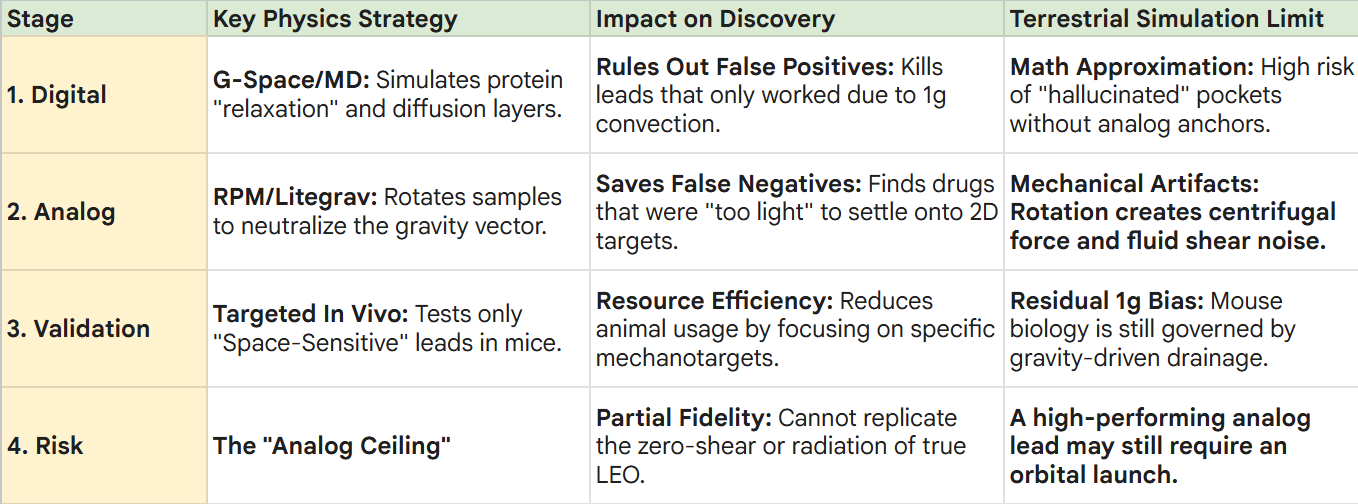
1. Digital Simulation (G-Space & Molecular Dynamics)
Before a sample enters a physical lab, researchers could use G-Space and in silico suites to predict “orbital feasibility.”
The Process:
Simulating the structural relaxation of proteins and the thick diffusion boundary layers of Low-Shear Modeled Microgravity (LSMMG).
Minimizing False Positives:
If the simulation shows that a drug’s “superiority” in 1g was actually caused by heat-driven convection (mixing) rather than actual binding affinity, the project is halted.
Terrestrial Strategies & Limits:
Strategy:
Utilizing Quantum Mechanics/Molecular Mechanics (QM/MM) to predict electron-level interactions in a “weightless” digital environment.
The Limit:
Computational models are only as good as their parameters. Without real-world analog data to “anchor” the simulation, digital models can drift into theoretical results that don’t translate to wet-lab biology.
2. Analog Validation (Random Positioning Machines)
Hypotheses that pass the digital sieve move to Random Positioning Machines (RPM) or high-speed clinostats like Litegrav.
The Process:
These devices rotate samples along multiple axes to neutralize the gravity vector, mimicking the “suspended” state of LEO.
The Consequence (Capturing False Negatives):
A drug that failed in 1g because it couldn’t “settle” onto a 2D surface might show a >2x improvement in an RPM-simulated 3D environment where it moves via pure diffusion.
This “saves” a potential breakthrough that would have been discarded in a conventional workflow.
Terrestrial Strategies & Limits:
Strategy:
Using Magnetic Levitation (MagLev) to suspend cells and simulate weightlessness.
The Limit:
RPMs and MagLev introduce Mechanical Artifacts. RPMs create centrifugal forces and fluid shear stress, while MagLev requires high-intensity magnetic fields that can interfere with cellular ion channels, potentially creating “false signals” that are purely magnetic, not gravitational.
3. Validation (Targeted Murine Models)
Only the “Space-Sensitive” candidates (those vetted by the digital and analog sieves) are moved into humanized mouse models.
The Process:
Standard 1g$ $in$ $vivo$ testing, but with a refined focus. Researchers look for the specific biomarkers (e.g., YAP/TAZ downregulation) that were identified during the RPM phase.
The Benefit: This targeted approach significantly reduces the number of animals needed. Instead of “spraying and praying” with 100 compounds, researchers move forward with 5 high-fidelity candidates that have already proven they can overcome mechanical resistance.
Terrestrial Strategies & Limits:
Strategy: Developing Micro-CT imaging to track drug penetration in live mice in real-time.
The Limit: The “Physics Mismatch” remains. Even a high-value candidate is being tested in a 1g animal. The mouse’s gravity-driven lymphatic system may still mask the drug’s true potential in a diffusion-dominated human tumor, leading to a lingering risk of failure at the clinical stage.
4. Workflow Risk (“The Strategic Middle”)
The Hybrid Workflow is a massive improvement over conventional methods, but it carries a Moderate Risk Profile:
Residual 1g Bias:
Because the final validation still occurs in 1g (animals or standard chips), the most subtle “pure space” effects (like large-scale protein crystal growth or scaffold-free tissue assembly) cannot be fully realized.
Complexity Overload:
Managing the data pipeline between G-Space simulations, RPM hardware, and animal labs requires a highly specialized team.
The “Analog Ceiling”:
While Litegrav minimizes gravitational noise, it cannot replicate the zero-shear and pure-vacuum/radiation conditions of LEO, meaning the “highest-fidelity” discovery might still be one launch away.
5. Clinical Readiness
In the Hybrid Workflow, “promising” results are characterized by a Mechanistic Signature. Unlike the conventional workflow, which relies on raw potency, the Hybrid path looks for proof that the drug’s success is tied to the physical state of the target.
A. The “Gravity-Neutral” Potency Spike
The most reliable indicator in a hybrid workflow is the Relative Improvement Metric (RIM) when transitioning from 1g to an analog environment.
The Result:
A drug shows modest efficacy in a standard incubator but a >2x increase in cell-killing or binding when placed in a Random Positioning Machine (RPM) or Litegrav device.
Clinical Significance:
This spike proves that the drug’s performance is hindered by gravity-induced settling or mechanical tension.
It suggests that in the “low-tension” environment of human soft tissue, the drug will perform significantly better than 1g lab tests initially suggested.
B. YAP/TAZ Pathway Normalization
The YAP/TAZ pathway is the cell’s “mechanical thermometer.” In 1g, these proteins are often over-activated due to the weight of the cell pressing against a plastic dish, driving drug resistance.
The Result:
The drug candidate successfully downregulates YAP/TAZ signaling specifically in the RPM-suspended state, whereas it failed to do so in the “flattened” 1g state.
Clinical Significance:
This provides a Mechanistic Biomarker. It proves the drug is a “Mechano-Inhibitor” capable of neutralizing the physical survival signals that cancers use to resist chemotherapy.
This is a high-confidence signal for Phase I trials in stiff, fibrotic tumors like Pancreatic Cancer.
C. The “Scaffold-Free” Penetration Coefficient
Traditional 1g tests use synthetic hydrogels (like Matrigel) that create artificial barriers. The Hybrid workflow uses analog microgravity to grow 3D spheroids without any support structures.
The Result:
The drug achieves deep penetration in a Scaffold-Free Spheroid grown in a clinostat, matching the diffusion rates predicted by G-Space simulations.
Clinical Significance:
This aligns Digital (Tier 1) and Analog (Tier 2) data. When the computer model and the RPM both show the same penetration speed, it provides a high-certainty “Physical Proof of Concept.”
It tells clinicians how the drug will behave in the fluid-filled voids of a human tumor, allowing for precise dosing.
While the Conventional Workflow proves a drug can kill cells in a dish, the Hybrid Workflow proves the drug can navigate the mechanical reality of a 3D human environment.
This reduces the risk of the “Phase II Cliff” by ensuring the drug’s mechanism isn’t just an artifact of a flat, plastic, 1g environment.
III. Microgravity-Maximal Workflow (Orbital Validation)
Focus: Utilizing Low Earth Orbit (LEO) as the primary “Operating System” for drug discovery, high-fidelity validation, and pilot-scale manufacturing of complex therapeutics.
1. In Silico (“Orbital Crystal” Benchmark)
Traditional modeling relies on 1g protein structures, which are physically “compressed” by gravity and cellular tension.
The Workflow:
Instead of guessing with terrestrial simulations, researchers grow high-purity protein crystals in LEO. These crystals are larger and more uniform because they grow without sedimentation or convection-driven defects.
The Consequence:
These “Orbital Crystals” reveal unmasked binding pockets; cryptic sites that are physically squeezed shut on Earth.
Molecular docking performed against these relaxed structures identifies high-affinity leads that 1g models would discard as “False Negatives.”
Space-Specific Limits & Strategies:
The Limit:
Growing crystals in space is a slow, time-dependent process; one “bad batch” can cost months of development time.
The Strategy:
Researchers use Automated Crystallization Platforms (e.g., Varda) that monitor growth via real-time telemetry.
If a crystal shows defects, the system can adjust the pH or temperature mid-flight to optimize the “perfect” structure for ground-based drug design.
2. In Vitro (Scaffold-Free “Macro-Organoids”)
Terrestrial 3D models are stunted by gravity, leading to flattened “pancakes” or models that require artificial scaffolds (like Matrigel) that skew drug penetration data.
The Workflow:
Large, patient-derived tumoroids are grown in a “suspended” liquid state in orbit.
Without gravity to collapse them, they self-organize into massive, perfectly spherical 3D architectures held together solely by their own natural Extracellular Matrix (ECM).
The Consequence:
These models possess a high-fidelity “Physical Shield” and a dense “Necrotic Core” that perfectly mirror human solid tumors.
Testing here provides a more accurate “Effective Diffusion Coefficient” for drug penetration.
Space-Specific Limits & Strategies:
The Limit:
Large organoids can become “too large,” suffering from nutrient deprivation at the center if the media isn’t perfectly managed.
The Strategy:
Scientists utilize Acoustic Levitators or Microfluidic Perfusion Loops in orbit. These systems gently circulate nutrients around the organoid without introducing the “fluid shear stress” that triggers false inflammatory signals on Earth.
3. In Vivo (“Systemic-on-a-Chip”)
Earth-bound mouse models have gravity-driven lymphatic and circulatory systems that don’t match human fluid dynamics, especially in the diffusion-dominated center of a tumor.
The Workflow:
Deployment of Longevity-Microenvironment-on-a-Chip (LMOC) platforms.
These chips connect multiple human organoids (e.g., Tumor + Bone Marrow + Liver) in a weightless circuit.
The Consequence:
Researchers track Immune Cell Trafficking (e.g., CAR-T cells) governed solely by chemical signals (chemotaxis), not gravity.
This isolates the “Pure Signal” of a T-cell’s ability to navigate to, breach, and kill a tumor mass.
Space-Specific Limits & Strategies:
The Limit:
Maintaining “living” chips in orbit for 30+ days requires complex life-support automation and radiation shielding.
The Strategy:
The use of Radiation-Hardened Bio-Capsules and AI-Driven Media Exchange.
These systems automatically monitor the health of the “systemic-on-a-chip” and transmit real-time metabolic data to ground teams, allowing for “Clinical-Mimic” dosing schedules.
4. Workflow Value (Solving the “Phase II Cliff”)
The Microgravity-Maximal workflow aims to reduce the False Positive trap of conventional research. By removing sedimentation and convection, drugs are tested against more similar physical constraints to what they will face inside a human patient:
True Interstitial Pressure: The drug must penetrate the tumor’s “shield” via pure affinity, not gravity.
Pure Diffusion Transport: Success is measured in the absence of heat-driven mixing (convection).
Natural Mechanical Resistance: The drug encounters a 360° spherical barrier, more precisely to what it exists in a human body.
The Result: A drug that passes the Microgravity-Maximal workflow enters Phase I trials with a stronger Data Package, having already survived a “physical stress test” that no Earth-based lab can currently replicate.
5. Clinical Readiness
Because LEO removes the confounding variables of sedimentation and convection, the resulting data package could potentially provide the highest possible “Probability of Success” (PoS) for human trials.
A. The “Pure Affinity” Binding Profile
On Earth, a drug might appear to bind well because it physically “settled” onto a cell. In LEO, binding is a choice made only by molecular recognition.
The Result:
A Binding Kinetic Curve that remains identical or improves in a zero-sedimentation environment.
Clinical Significance:
This proves “Pure Molecular Affinity.” It tells the FDA that the drug’s efficacy is driven by its chemical design, not by gravitational luck.
This is a key “de-risking” metric for high-potency drugs like radiopharmaceuticals or antibody-drug conjugates (ADCs) where off-target binding means high toxicity.
B. The “Macro-Organoid” Kill Zone
Because LEO allows for the growth of massive, scaffold-free tumoroids, researchers can measure the drug’s ability to reach the “Necrotic Core”; the oxygen-starved center where the most resistant cancer cells hide.
The Result:
High-resolution microscopy showing Total Volume Penetration (TVP).
Success is defined by the drug reaching the center of a 5mm spherical mass without the aid of artificial convection.
Clinical Significance:
If a drug can kill the core of an orbital macro-organoid, it has survived the “Physical Stress Test” of a real human tumor.
This is a strong predictor of whether a drug will succeed in a Phase II trial for “hard-to-treat” cancers like glioblastoma or pancreatic ductal adenocarcinoma.
C. Systemic “Inter-Organ” Signaling
Utilizing the Longevity-Microenvironment-on-a-Chip (LMOC), researchers can observe how a drug affects not just the tumor, but the connected immune and metabolic systems.
The Result:
A Cross-Talk Fingerprint that shows the drug killing the tumor while simultaneously rejuvenating or protecting connected bone marrow or liver organoids.
Clinical Significance:
This provides a Multi-Organ Safety Profile.
It mimics the systemic complexity of a human patient better than a mouse model.
If the “bone marrow-on-a-chip” remains healthy while the tumor dies in a weightless circuit, the drug can have a higher probability for being cleared for human trials with a significantly lower risk of systemic side effects.