Oncology Simulated Microgravity Experiments [Strategy]
With orbital launch slots at a premium, a strategic priority is on Pre-Flight De-Risking.
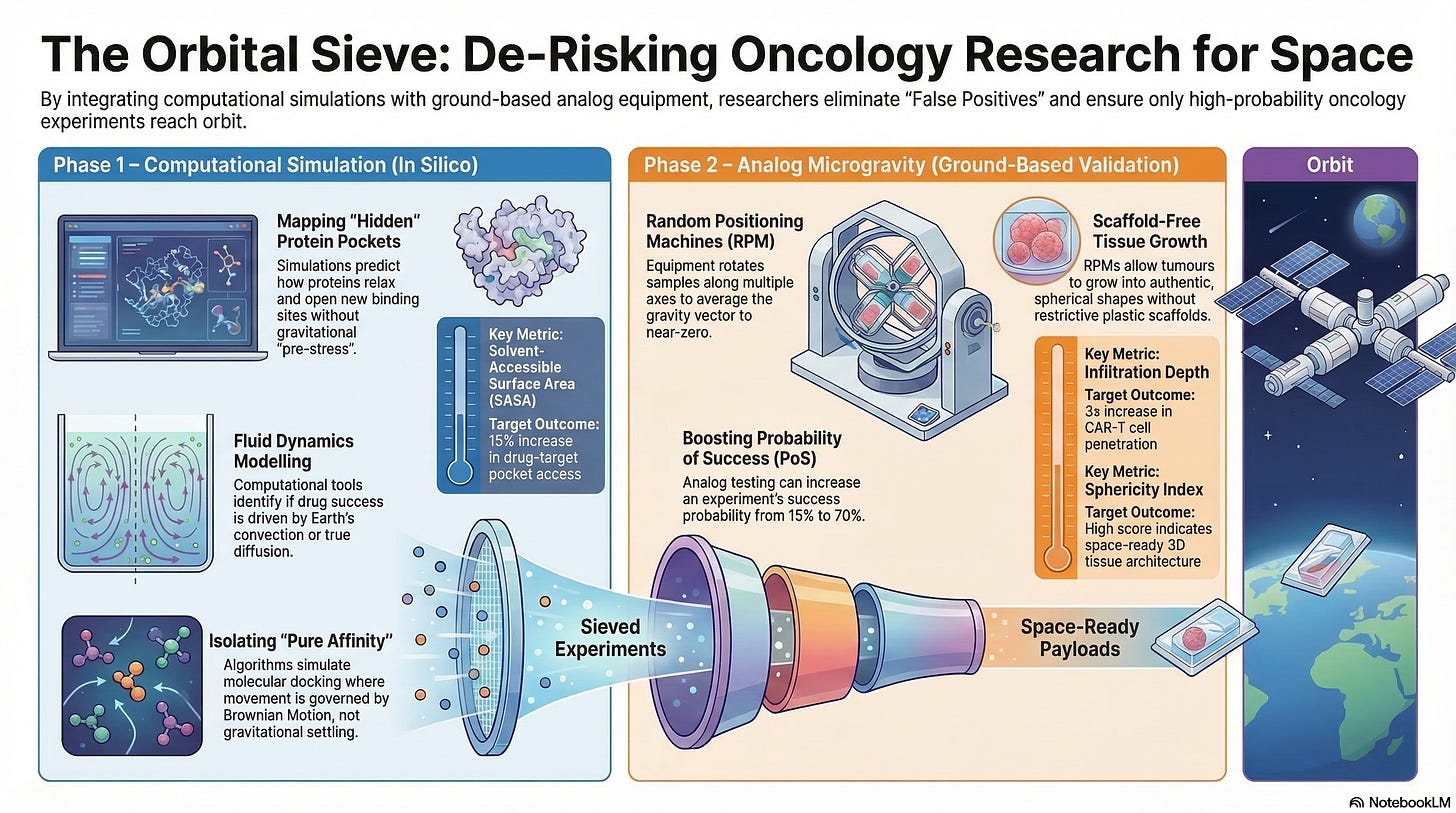
With orbital launch slots at a premium, a strategic priority is on Pre-Flight De-Risking. A goal is to ensure that every milligram of payload has been vetted through a rigorous computational and analog sieve.
By integrating Computational Biology (e.g., G-Space) with Ground-Based Analog Equipment (e.g., Litegrav), oncology researchers can attempt to effectively minimize “False Negatives” (missing a space-only discovery) and “False Positives” (launching experiments that could have been solved on Earth).
I. Computational Simulation (In Silico Modeling)
Tools like G-Space, AlphaFold-Sim, and advanced Molecular Dynamics (MD) suites allow researchers to create a “Digital Twin” of the microgravity environment.
This computational layer acts as the first filter in the “Orbital Sieve,” specifically targeting the physics of the Low-Shear Modeled Microgravity (LSMMG) environment to predict biological outcomes before physical resources are committed.
1. Structural Biology (Mapping the “Mechanosensitive” Proteome)
On Earth, gravity exerts a constant “pre-stress” on the cellular cytoskeleton (tensegrity). This tension physically tugs on proteins, often keeping certain binding pockets closed or “masked.”
Identifying “Hidden Pockets”:
Computational tools simulate the Unloading Event.
By virtually removing the 9.81 m/s^2 vector, MD simulations can predict how a protein’s 3D conformation “relaxes.”
The “Pocket-Opening” Hypothesis:
Simulations might reveal a cryptic binding site on a signaling protein (like KRAS or MYC) that only becomes accessible when the cytoskeletal “tethers” are relaxed.
Maximizing True Positives:
If the simulation shows a 15% increase in the solvent-accessible surface area (SASA) of a drug-target pocket in microgravity, that molecule could become a Tier-1 Launch Candidate.
This could prevent “False Negatives”; missing a drug that only works when the target is physically relaxed.
2. Fluid Dynamics (Predicting the “Diffusion-Only” Zone)
One of the most significant differences in LEO is the near-total elimination of Gravity-Driven Convection. On Earth, temperature differences cause fluids to move (convection), which can mask how a drug actually diffuses into a cell.
The LSMMG Simulation:
Computational tools model the fluid boundary layer around a 3D tumoroid.
In microgravity, this layer is much thicker, creating a “Diffusion-Limited” environment.
Minimizing False Positives:
If a researcher hypothesizes that a drug will penetrate a tumor better in space, but the Computational Fluid Dynamics (CFD) model shows that the lack of convection actually slows the drug’s arrival at the cell surface, the hypothesis is flagged as a likely False Positive.
The “Rule-Out”:
If the 1g convection is what was actually driving the drug’s “success” in the lab, the space experiment would fail.
3. Ligand-Receptor Docking (Isolating “Pure Affinity”)
On Earth, the “Targeting Specificity” of a nanoparticle or antibody is often skewed by Sedimentation. Large molecules “fall” onto cells, creating “forced” interactions that wouldn’t happen in a weightless environment.
Virtual Docking Sieve:
Algorithms simulate docking kinetics where the movement of the drug is governed solely by Brownian Motion rather than gravitational settling.
Data Point: Binding Energy
If the simulation predicts that the Binding Energy of a nanoparticle is the same in both 1g and microgravity, then the observed “benefit” of space is likely zero.
Strategy:
Only prioritize ligands where the simulation shows a Binding Kinetic Advantage in a non-sedimenting environment.
This ensures that we are launching “Pure Affinity” experiments that rely on molecular recognition, not gravitational luck.
4. Mechanotransduction (The YAP/TAZ Pathway Simulation)
The YAP/TAZ pathway is the cell’s primary “gravity sensor.” When gravity is removed, the signals sent from the cell membrane to the nucleus change instantly.
The Experiment:
Before launching a “Longevity” or “Anti-Cancer” drug that targets these pathways, researchers could run a Systems Biology Simulation.
Rule-Out Strategy:
If the computational model predicts that the cell will “compensate” for the lack of gravity within 48 hours by upregulating alternative tension pathways (like RhoA), then a short-duration (3-day) mission is ruled out as likely to yield a “False Positive” result that won’t last.
Strategy:
This data dictates Mission Duration.
If the simulation shows the “Rejuvenation Signal” peaks at day 14, the researcher knows to book a long-duration slot on a commercial station (like Axiom or Orbital Reef) rather than a short sub-orbital flight.
II. Analog Microgravity (Random Positioning Machines)
Analog Microgravity can serve as the critical “wet-lab” bridge between digital simulations and Space Stations. Devices like High-Performance Random Positioning Machines (RPM) and Clinostats (such as the Litegrav system) do not eliminate gravity, but they rotate a biological sample along multiple axes so that the gravity vector is averaged to near-zero over time.
This state, known as Modeled Microgravity, is a possible “Stress-Test” for oncology and longevity hypotheses.
By the time a payload reaches a Varda or SpaceX capsule, the Analog Microgravity phase could increase the “Probability of Success” (PoS) from a speculative 15% to a robust 70%.
The RPM can de-risks the capital investment of orbital manufacturing. It ensures that when we finally “turn off” gravity in LEO, we are doing so to confirm a breakthrough that we have already seen whispering in the rotation of an analog machine on Earth.
1. Functional Validation (The “Biological Signal” Check)
Once a computational model predicts a “hidden pocket” or an “affinity advantage,” the RPM could be used to see if biology actually cooperates. This stage is useful for Minimizing False Negatives; detecting breakthroughs that are physically impossible to see in a standard 1g incubator.
The “Sedimentation-Free” Zone:
On Earth, 3D tumoroids settle at the bottom of a flask, leading to unnatural nutrient gradients and physical compression.
In an RPM, the tumoroid remains suspended in the media.
Case Study: CAR-T Infiltration.
A researcher might find that a specific CAR-T therapy has mediocre results in 1g.
However, when placed in an RPM-simulated 3D environment, the “buoyancy” of the cells allows them to navigate the tumoroid’s periphery without being pinned down by gravity.
The Signal:
If the RPM shows a 3x increase in Mean Infiltration Depth (MID), the researcher has found a “Space-Specific Benefit.” Without this analog step, the therapy might have been discarded as a failure, a classic False Negative.
2. Identifying “Analog Noise” vs. “Orbital Signal”
While RPMs are powerful, they introduce their own forces, specifically Centrifugal Force and Fluid Shear Stress. Part of the strategy is using high-performance analogs to separate these artifacts from true microgravity effects.
The “Shear Sieve”:
If a drug only works when the RPM is spinning at high speeds, the effect might be due to fluid “mixing” (shear) rather than weightlessness.
Strategy:
Researchers run a Speed-Gradient Test.
If the biological benefit (e.g., tumor cell death) disappears at lower rotation speeds, the hypothesis is likely a False Positive for space; it’s just a reaction to fluid movement.
The “Pure” Candidate:
A true orbital candidate shows consistent results across different analog rotation profiles, suggesting the effect is driven by the Neutralization of the Gravity Vector itself.
3. Morphological “Pre-Evolution” (Growing Larger Organoids)
One of the highest-value uses of Litegrav-style equipment is the preparation of “Space-Ready” tissues. High-fidelity organoids take time to mature, and doing the initial growth on the ground can be cost-effective.
Scaffold-Free Growth:
In 1g, large organoids require plastic scaffolds to keep them from collapsing under their own weight.
These scaffolds interfere with drug diffusion.
Analog Advantage:
An RPM allows for Scaffold-Free maturation.
The tissue grows into a more authentic, spherical shape held together only by its own Extracellular Matrix (ECM).
The Data: Sphericity Index
Researchers measure how close the organoid is to a perfect sphere.
A high $Sphericity Index in the analog is a prerequisite for launch, as it ensures the 3D architecture is ready to replicate the “necrotic core” dynamics found in real human tumors.
III. Example Oncology Strategies (Ruling Out vs. Doubling Down)
Below are five example case studies applying this tiered de-risking strategy to high-value oncology therapy types.
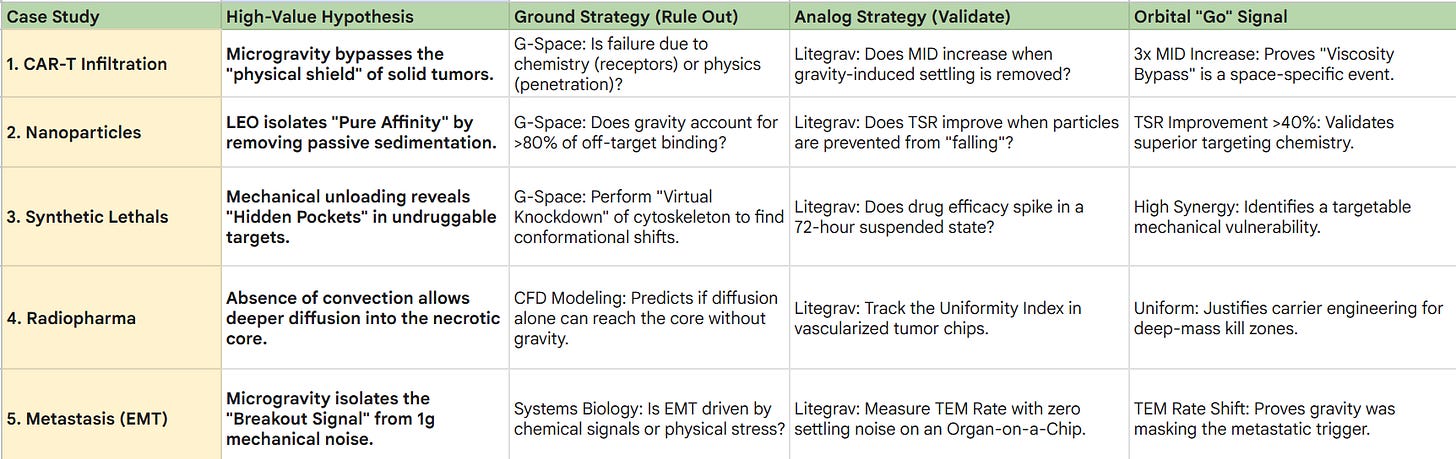
1. CAR-T Infiltration (The “Viscosity Bypass”)
The Hypothesis:
Microgravity allows CAR-T cells to navigate the dense stroma of Pancreatic Duct Adenocarcinoma (PDAC) more effectively by neutralizing the gravity-induced settling that “pins” cells to the tumor periphery.
Ground Strategy (Rule Out):
Use G-Space to model the physical “Stroma-Wall.”
If the simulation shows that the drug’s failure in 1g is purely biochemical (e.g., poor receptor matching) rather than physical (e.g., poor penetration depth), the hypothesis is ruled out to save launch costs.
Analog Strategy (Validate):
Grow PDAC tumoroids in a Litegrav RPM.
If the CAR-T cells show zero improvement in Mean Infiltration Depth (MID) compared to 1g controls, the theory is a False Positive.
The Pivot:
If Litegrav reveals that CAR-T cells “drill” significantly deeper only when suspended, you have a high-probability orbital candidate. Data on the Exhaustion Profile from the RPM informs whether to add checkpoint inhibitors to the flight payload.
2. Nanoparticle “Pure Affinity” Testing
The Hypothesis:
Targeting TNBC nanoparticles settle onto healthy cells on Earth due to passive sedimentation.
In LEO, they will bind only via active ligand-receptor docking.
Ground Strategy (Minimizing False Negatives):
Computational tools analyze the Sedimentation Rate vs. Binding Energy.
If the software predicts that gravity is responsible for >80% of off-target binding, the project is fast-tracked as “High-Yield” for orbit.
Analog Strategy:
Test the nanoparticle in a Litegrav clinostat to neutralize settling.
The Data: Targeting Specificity Ratio (TSR).
If Litegrav improves the TSR by >40%, the targeting chemistry is inherently superior and ready for launch.
If TSR remains low, the toxicity is chemical, and the drug must be reformulated before any orbital mission is considered.
3. Precision “Synthetic Lethal” Targets
The Hypothesis:
“Undruggable” mutations (e.g., KRAS) become vulnerable when the cell’s internal skeleton (cytoskeleton) is “relaxed” in microgravity, revealing hidden binding pockets.
Ground Strategy:
Use G-Space to perform a “Virtual Knockdown” of cytoskeletal proteins like Actin.
If the simulation doesn’t show a conformational change in the target protein, the mission is canceled.
Analog Strategy:
Maintain cells in a Litegrav RPM for 72 hours to simulate mechanical unloading.
The Data: Synergy Coefficient (α).
If the drug’s efficacy spikes in the RPM state, you have identified a Mechanical Vulnerability.
This allows the company to launch a “Space-Hardened” lead compound to find binding sites that will eventually be targeted on Earth using mechano-mimetic drugs.
4. Radiopharmaceuticals (3D Dose Optimization)
The Hypothesis:
Radioligands fail in 1g because they cannot reach the “necrotic core” of a tumor due to gravity-driven fluid stagnation.
Ground Strategy:
Use CFD (Computational Fluid Dynamics) to simulate the Diffusion-Only Zone of a vascularized tumor chip.
If the model predicts that diffusion alone is insufficient to reach the core even without gravity, the carrier molecule is too large and must be redesigned.
Analog Strategy:
Deploy vascularized chips in an RPM.
Track the Uniformity Index (Ui) of radiation absorption across the spheroid.
The Strategic Move:
If Litegrav shows a Ui improvement (more uniform spread), the hypothesis is doubled down for LEO.
This data informs Carrier Engineering, switching to smaller, higher-diffusivity peptide carriers before the flight to maximize the “kill zone.”
5. Metastasis & EMT (Mapping the “Breakout” Signal)
The Hypothesis:
The “breakaway” signal that turns a primary tumor cell into a metastatic killer is triggered by fluid shear stresses that are confounded by gravity on Earth.
Ground Strategy:
Use Systems Biology modeling to identify genes upregulated by “Mechanical Stress.”
If the model suggests the EMT transition is purely chemical (TGF-beta driven) and not mechanical, the orbital study of “breakout signals” is deprioritized.
Analog Strategy:
Utilize an “Organ-on-a-Chip” in a Litegrav RPM to monitor cell detachment with zero settling noise.
The Data: Trans-Endothelial Migration (TEM) Rate.
If the TEM rate changes significantly in the analog environment, it proves gravity was masking the “breakout” signal.
Lead Selection:
The drug that best reduces the TEM rate in Litegrav is prioritized for the flight mission to identify the specific RNA “exit signals” used as companion diagnostics for Phase I trials.